
(a)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
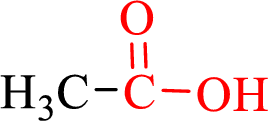
Nomenclature of
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the
alkane name with –oic acid.
Naming of compounds with two
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
Carboxylic salts are the water-soluble ammonium or alkali metal salts of carboxylic acids.
(a)
Explanation of Solution
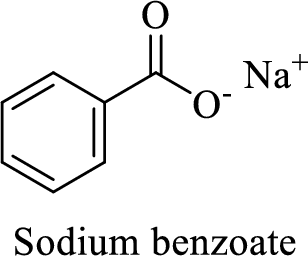
Name of the given salt is sodium benzoate.
From the name, we will get the following fact about the structure of the compound.
- ✓ The given compound is a sodium salt of benzoic acid.
Thus,
The structural formula for this salt can be drawn as shown below,
(b)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
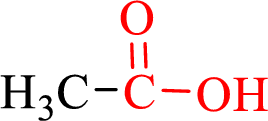
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid. If two or more carboxylic functional groups are present in the same compound then its number should be taken in to consideration and the prefix di, tri, tetra.. must be used.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
Carboxylic salts are the water-soluble ammonium or alkali metal salts of carboxylic acids.
(b)
Explanation of Solution
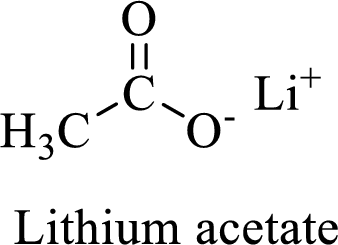
Name of the given salt is Lithium acetate.
From the name, we will get the following fact about the structure of the compound.
- ✓ The given compound is a Lithium salt of acetic acid
Thus,
The structural formula for this compound can be drawn as shown below,
(c)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
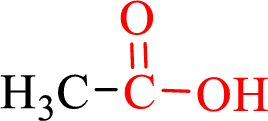
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
Carboxylic salts are the water-soluble ammonium or alkali metal salts of carboxylic acids.
(c)
Explanation of Solution
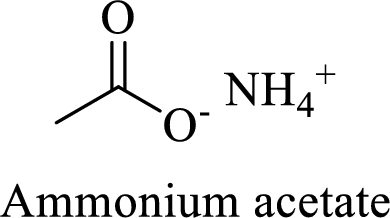
Name of the given salt is Ammonium acetate.
From the name, we will get the following fact about the structure of the compound.
- ✓ The given compound is an ammonium salt of acetic acid.
Thus,
The structural formula for this compound can be drawn as shown below,
(d)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
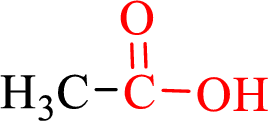
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
Carboxylic salts are the water-soluble ammonium or alkali metal salts of carboxylic acids.
(d)
Explanation of Solution
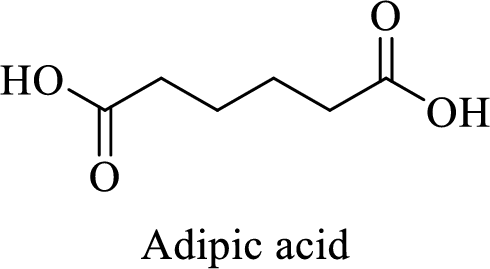
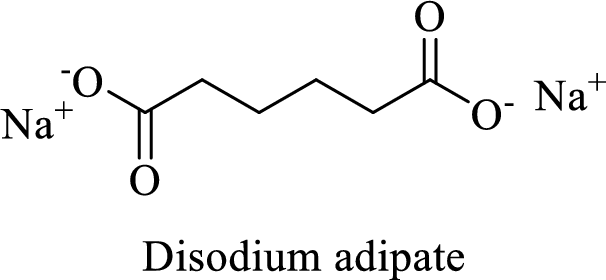
Name of the given salt is Disodium adipate.
From the name, we will get the following facts about the structure of the compound.
- ✓ The given compound is a sodium salt of adipic acid.
- ✓ Two sodium atoms are attached to the both carboxyl group of adipic acid.
The structure of adipic acid is shown below,
Thus,
The structural formula for this compound can be drawn as shown below,
(e)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
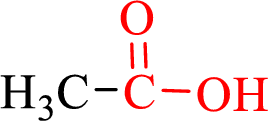
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
Carboxylic salts are the water-soluble ammonium or alkali metal salts of carboxylic acids.
(e)
Explanation of Solution
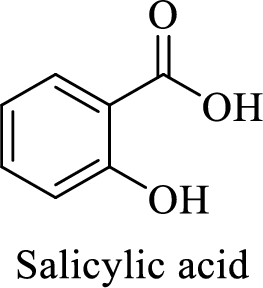
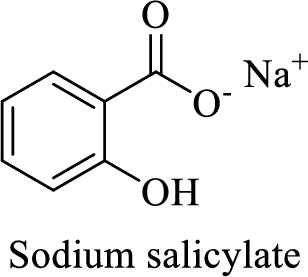
Name of the given salt is sodium salicylate.
From the name, we will get the following fact about the structure of the compound.
- ✓ The compound is a sodium salt of salicylic acid.
Structure of salicylic acid is shown below,
Thus,
The structural formula for this compound can be drawn as shown below,
(f)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
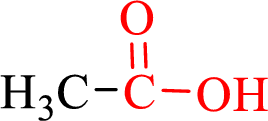
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid. If two or more carboxylic functional groups are present in the same compound then its number should be taken in to consideration and the prefix di, tri, tetra.. must be used.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
Carboxylic salts are the water-soluble ammonium or alkali metal salts of carboxylic acids.
(f)
Explanation of Solution
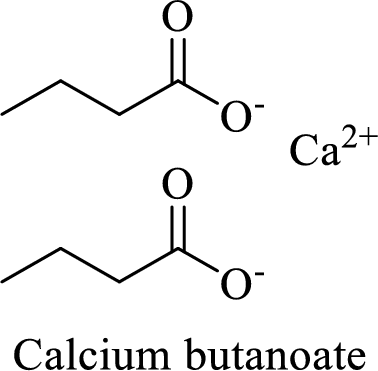
Name of the given salt is calcium butanoate.
From the name, we will get the following fact about the structure of the compound.
- ✓ The given compound is a calcium salt of butanoic acid.
Thus,
The structural formula for this compound can be drawn as shown below,
Want to see more full solutions like this?
Chapter 17 Solutions
Organic Chemistry
- Now that you have learned about solubility, explain why aspirin crosses a cell membrane as a neutral carboxylic acid rather than an ionic conjugate base ?arrow_forwardWhat is the role of phenolphthalein in the neutralization reaction? Draw the structure of phenolphthalein under acidic and basic conditions.arrow_forwardWhat phase is Benzoic acid, benzil, alizarin, ethyl acetate, and aqueous NaOH at in room temperature? solid, liquid or solution?arrow_forward
- Greetings, Can you please provide the solution to the attached question. Thank youarrow_forwardIs the sodium salt of propanoic acid as effective a soap assodium stearate? Explain.arrow_forwardHello, I'm having trouble understanding the solution and couldn't make out the handwriting of the expert. Thank you in advance.arrow_forward
- Plese don't provide handwritten solition..arrow_forwardWhat is the theoretical COD of mixture containing 150 mg/L phenol and 50 mg/L acetic acidarrow_forwardThe compound acetophenone has a very similar molar mass to that of benzoic acid and benzamide. However, acetophenone has a much lower m.p. (20 °C) than both such that, by contrast, it is a liquid at room temperature. By considering intermolecular forces and comparing functional group structure, account for this big difference in physical properties.arrow_forward
- C7 Please help me with the brief solution and answer, thank youarrow_forwardExplain how you would separate a mixture containing benzoic acid and a neutral substance, benzophenone. Both compounds are insoluble in water and soluble in ether. *Do not send flow charts. Please explainarrow_forwardWhy are the acidic and basic extracts cooled before neutralization?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





