
(a)
Interpretation:
Whether the given statement is true or false.
Keto and enol tautomers are constitutional isomers.
Concept Introduction:
A carbonyl compound that has a hydrogen on an a-carbon changes its form to enol through resonance. Both the form stays in equilibrium.
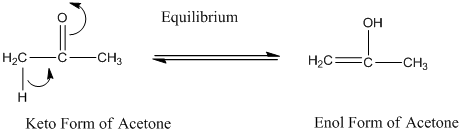
Constitutional isomers are those where the molecular formula for both the compound is same but the bond connectivity differs.
(b)
Interpretation:
Whether the given statement is true or false.
For a pair of keto-enol tautomers, the keto form generally predominates.
Concept Introduction:
A carbonyl compound that has a hydrogen on an a-carbon changes its form to enol through resonance. Both the form stays in equilibrium.
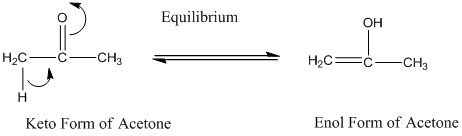
Constitutional isomers are those where the molecular formula for both the compound is same but the bond connectivity differs.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Introduction to General, Organic and Biochemistry
- 17-47 What is the characteristic structural feature of a hemiacetal? Of an acetal?arrow_forward17-34 Explain why liquid aldehydes are often stored under an atmosphere of nitrogen rather than in air.arrow_forward17-26 Account for the fact that acetone has a higher boiling point (56°C) than ethyl methyl ether (11°C) even though their molecular weights are almost the same.arrow_forward
- 17-11 What is the difference in structure between an aromatic aldehyde and an aliphatic aldehyde?arrow_forward17-70 What simple chemical test could you use to distinguish between the members of each pair of com pounds? Tell what you would do, what you would expect to observe, and how you would interpret your experimental observation. (a) Benzaldehyde and cyclohexanone (b) Acetaldehyde and acetonearrow_forward16-28 Following is the structural formula of metformin, the hydrochloride salt of which is marketed as the antidiabetic medication Glucophage. Metformin was introduced into clinical practice in the United States in 1995 for the treatment of type 2 diabetes. More than 25 million prescriptions for this drug were written in 2000, making it the most commonly prescribed brand-name diabetes medication in the nation. NH NH H3(\ 3 N N Nh2ch3 h Metformin Complete the Lewis structure for metformin, showing all valence electrons. Which nitrogen is the most likely site of protonation? Draw the structural formula of Glucophage.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
