
Concept explainers
(a)
Interpretation:
The product formed when Propanal reacts with Methanol.
Concept Introduction:
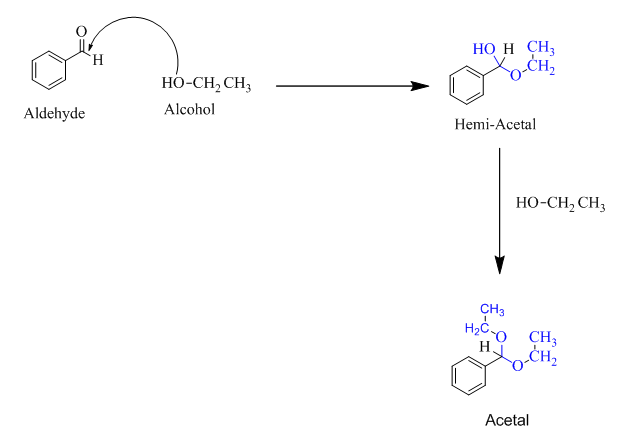
Hemiacetal is a type of molecule which contains carbon bonded to one -OH group and one -OR group. It is basically a half acetal. A hemiacetal forms when one molecule of alcohol reacts with a carbonyl group of an
For an Example:
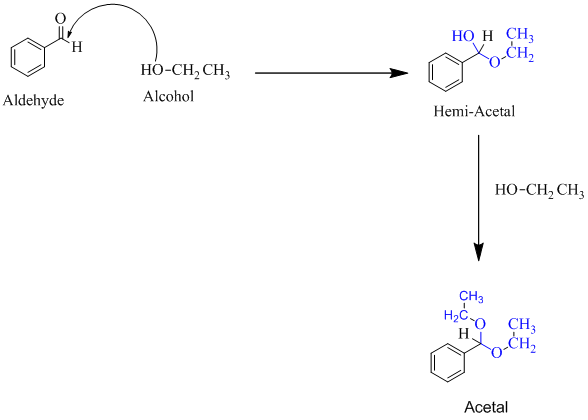
(b)
Interpretation:
The product formed when Cyclopentanone reacts with Methanol.
Concept Introduction:
Hemiacetal is a type of molecule which contains carbon bonded to one -OH group and one -OR group. It is basically a half acetal. A hemiacetal forms when one molecule of alcohol reacts with a carbonyl group of an aldehyde or ketone.
For an Example:
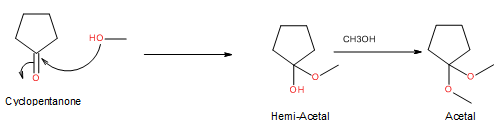
Trending nowThis is a popular solution!

Chapter 17 Solutions
Introduction to General, Organic and Biochemistry
- 17-70 What simple chemical test could you use to distinguish between the members of each pair of com pounds? Tell what you would do, what you would expect to observe, and how you would interpret your experimental observation. (a) Benzaldehyde and cyclohexanone (b) Acetaldehyde and acetonearrow_forward17-69 Propanal (bp 49°C) and 1-propanol (bp 97°C) have about the same molecular weight, yet their boiling points differ by almost 50°C. Explain this fact.arrow_forward17-33 What simple chemical test could you use to distinguish between the members of each pair of com pounds? Tell what you would do, what you would expect to observe, and how you would interpret your experimental observation. (a) Pentanal and 2-pentanone (b) 2-Pentanone and 2-pentanolarrow_forward
- 17-26 Account for the fact that acetone has a higher boiling point (56°C) than ethyl methyl ether (11°C) even though their molecular weights are almost the same.arrow_forward16-28 Following is the structural formula of metformin, the hydrochloride salt of which is marketed as the antidiabetic medication Glucophage. Metformin was introduced into clinical practice in the United States in 1995 for the treatment of type 2 diabetes. More than 25 million prescriptions for this drug were written in 2000, making it the most commonly prescribed brand-name diabetes medication in the nation. NH NH H3(\ 3 N N Nh2ch3 h Metformin Complete the Lewis structure for metformin, showing all valence electrons. Which nitrogen is the most likely site of protonation? Draw the structural formula of Glucophage.arrow_forward17-3 1 Draw a structural formula for the principal organic product formed when each compound is treated with K2Cr2O7/H2SO4. If there is no reaction, say so. (a) Butanal (b) Benzaldehyde (c) Cyclohexanone (d) Cyclohexanolarrow_forward
- 17-27 Pentane, 1-butanol, and butanal all have approximately the same molecular weights but different boiling points. Arrange them in order of increasing boiling point. Explain the basis for your ranking.arrow_forward17-35 Suppose that you take a bottle of benzaldehyde (a liquid, bp 179°C) from a shelf and find a white solid in the bottom of the bottle. The solid turns litmus red; that is, it is acidic. Yet aldehydes are neutral compounds. How can you explain these observations?arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
