
Concept explainers
17-54 Following is the structure of immunosuppressant FK-506, a molecule shown to disrupt calcineurin-mediated signal transduction in T-lymphocytes.
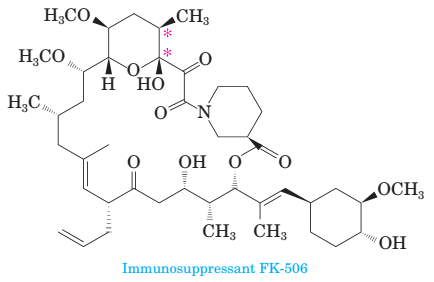
(a) There are three carbon—carbon double bonds present in this molecule. Which of the three has the potential for cis/trans isomerism? Assign a cis or trans con?guration to each carbon-carbon double bond that has this possibility.
(b) How many stereocenters are present in this molecule? How many stereoisomers are possible for it?
(c) Are there any aromatic components in this molecule?
(d) Consider the two carbon atoms marked with asterisks. Assign an R or S con?guration of each stereocenter.
(e) Because of the presence of a 21-member ring, this molecule is described as a macrocycle. This ring is fashioned by three types of bonds, several carbon-carbon bonds, one ester, one hemiacetal, and one amide. Locate the ester and the hemiacetal.
(f) Draw the structural formula of the long chain compound that would result if the hemiacetal were to be cleaved to an alcohol and a carbonyl group.
Trending nowThis is a popular solution!

Chapter 17 Solutions
Introduction to General, Organic and Biochemistry
- 17-74 Glucose, C6H12O6, contains an aldehyde group but exists predominantly in the form of the cyclic hemiacetal shown here. We will discuss this cyclic form of glucose in Chapter 20. A cyclic hemiacetal is formed when the —OH group of one carbon bonds to the carbonyl group of another carbon. (a) Which carbon in glucose provides the —OH group and which provides the —CHO group? (b) Draw the alternative chair confirmations of D-glucose and state which of the two is the more stable.arrow_forward16-28 Following is the structural formula of metformin, the hydrochloride salt of which is marketed as the antidiabetic medication Glucophage. Metformin was introduced into clinical practice in the United States in 1995 for the treatment of type 2 diabetes. More than 25 million prescriptions for this drug were written in 2000, making it the most commonly prescribed brand-name diabetes medication in the nation. NH NH H3(\ 3 N N Nh2ch3 h Metformin Complete the Lewis structure for metformin, showing all valence electrons. Which nitrogen is the most likely site of protonation? Draw the structural formula of Glucophage.arrow_forward13-49 (Chemical Connections 13F) In what ways is capsaicin used in medicine?arrow_forward
- 13-31 What structural features are common to vitamin E, BHT, and BHA (the three antioxidants presented in Section 13-40?arrow_forward14-74 Following is a structural formula for the amino acid cysteine: O II HS—CH — CH—C —OH I nh2 Name the three functional groups in cysteine. In the human body, cysteine is oxidized to cystine, a disulfide. Draw a structural formula for cystine.arrow_forward13-52 2,6-Di-/ezY-butyl-4-methylphenol (BHT, Section 13-4C) is an antioxidant added to processed foods to “retard spoilage.” How does BHT accomplish this goal?arrow_forward
- 13-27 Define autoxidation.arrow_forward16-17 Propylamine (bp 48°C), ethylmethylamine (bp 37°C), and trimethylamine (bp 3°C) are constitutional isomers with the molecular formula C3HgN. Account for the fact that trimethylamine has the lowest boiling point of the three and propylamine has the highest.arrow_forward16-54 Several poisonous plants, including Atropa belladonna, contain the alkaloid atropine. The name “belladonna” (which means “beautiful lady”) probably comes from the fact that Roman women used extracts from this plant to make themselves more attractive. Atropine is widely used by ophthal mologists and optometrists to dilate the pupils for eye examination. Classify the amino group in atropine as primary, secondary, or tertiary. Locate all stereocenters in atropine. Account for the fact that atropine is almost insoluble in water (1 g in 455 mL of cold water) but atropine hydrogen sulfate is very soluble (1 g in 5 mL of cold water). Account for the fact that a dilute aqueous solution of atropine is basic (pH approximately 10.0).arrow_forward
- 14-51 (Chemical Connections 14B) When was nitroglycerin discovered? Is this substance a solid, a liquid, or a gas'arrow_forward17-12 Is it possible for the carbon atom of a carbonyl group to be a stereocenter? Explain.arrow_forward13-38 (Chemical Connections 13A) What is meant by the term biodegradable?arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
