
(a)
Interpretation:
Form the given molecules; we need to find out the compound which reduced in presence of a reducing agent to give the given product.
Concept Introduction:
Generally an
Mechanism for NaBH4 reduction explained below.
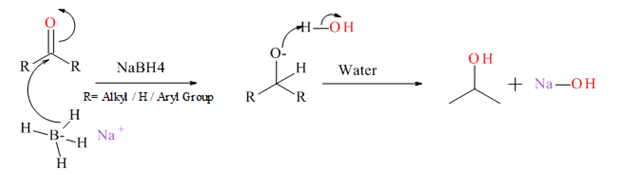
(b)
Interpretation:
Form the given molecules; we need to find out the compound which reduced in presence of a reducing agent to give the given product.
Concept Introduction:
Generally an aldehyde or ketone reduced to give the corresponding 1° or 2° alcohol. There is several type of reducing agents such as Pd /H2, NaBH4, LiAlH4 etc.
Mechanism for NaBH4 reduction explained below.
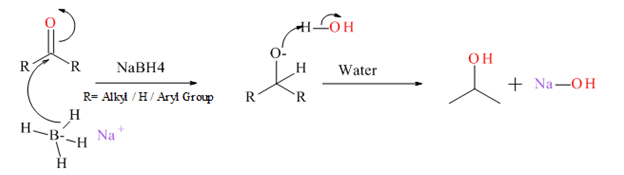
(c)
Interpretation:
Form the given molecules; we need to find out the compound which reduced in presence of a reducing agent to give the given product.
Concept Introduction:
Generally an aldehyde or ketone reduced to give the corresponding 1° or 2° alcohol. There are several type of reducing agents such as Pd /H2, NaBH4, LiAlH4 etc.
Mechanism for NaBH4 reduction explained below.
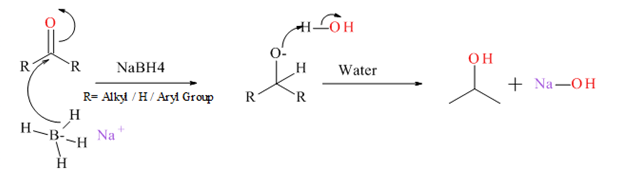
(d)
Interpretation:
Form the given molecules; we need to find out the compound which reduced in presence of a reducing agent to give the given product.
Concept Introduction:
Generally an aldehyde or ketone reduced to give the corresponding 1° or 2° alcohol. There are several type of reducing agents such as Pd /H2, NaBH4, LiAlH4 etc.
Mechanism for NaBH4 reduction explained below.
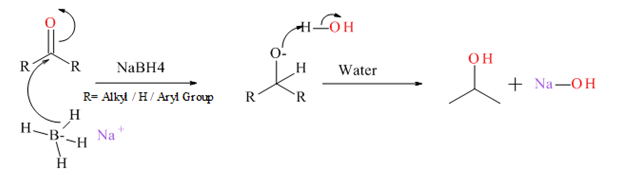
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Introduction to General, Organic and Biochemistry
- 17-70 What simple chemical test could you use to distinguish between the members of each pair of com pounds? Tell what you would do, what you would expect to observe, and how you would interpret your experimental observation. (a) Benzaldehyde and cyclohexanone (b) Acetaldehyde and acetonearrow_forward17-11 What is the difference in structure between an aromatic aldehyde and an aliphatic aldehyde?arrow_forward17-47 What is the characteristic structural feature of a hemiacetal? Of an acetal?arrow_forward
- 17-28 Show how acetaldehyde can form hydrogen bonds with water.arrow_forward17-13 Which compounds contain carbonyl groups?arrow_forward17-72 The following molecule is an enediol; each carbon of the double bond carries an —OH group. Draw structural formulas for the hydroxyketone and the a-hydroxyaldehyde with which this enediol is in equilibrium.arrow_forward
- 17-60 1-Propanol can be prepared by the reduction of an aldehyde, but it cannot be prepared by the acid catalyzed hydration of an alkene. Explain why it cannot be prepared from an alkene.arrow_forward18-41 Complete these examples of Fischer esterification. In each case, assume an excess of the alcohol.arrow_forward14-55 (Chemical Connections 140 The legal definition of being under the influence of alcohol is based on blood alcohol content. What is the relationship between breath alcohol content and blood alcohol content?arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
