
(a)
Interpretation:
Draw the structural formula the compound formed by the reaction of given compound with sodium borohydride.
Concept Introduction:
Answer to Problem 17.65P
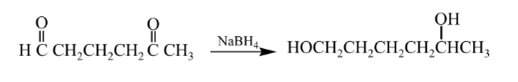
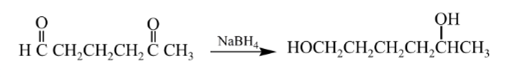
Explanation of Solution
Aldehydes are reduced to primary alcohols and ketones are reduced in secondary alcohols. The most commonly used reagent for reduction of aldehydes and ketone is sodium borohydride
Therefore, the product formed will be as follows:
(b)
Interpretation:
Draw the structural formula the compound formed by the reaction of given compound with sodium borohydride.
Concept Introduction:
Aldehydes are reduced to primary alcohols and ketones are reduced in secondary alcohols. The most commonly used reagent for reduction of aldehydes and ketone is sodium borohydride
Answer to Problem 17.65P
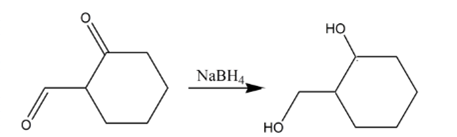
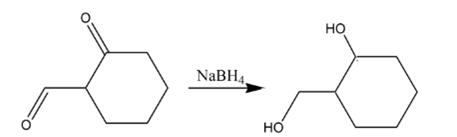
Explanation of Solution
Aldehydes are reduced to primary alcohols and ketones are reduced in secondary alcohols. The most commonly used reagent for reduction of aldehydes and ketone is sodium borohydride
Therefore, the product formed will be as follows:
(c)
Interpretation:
Draw the structural formula the compound formed by the reaction of given compound with sodium borohydride.
Concept Introduction:
Aldehydes are reduced to primary alcohols and ketones are reduced in secondary alcohols. The most commonly used reagent for reduction of aldehydes and ketone is sodium borohydride
Answer to Problem 17.65P
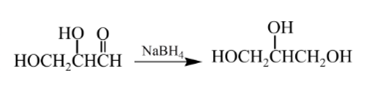
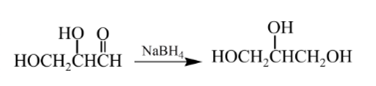
Explanation of Solution
Aldehydes are reduced to primary alcohols and ketones are reduced in secondary alcohols. The most commonly used reagent for reduction of aldehydes and ketone is sodium borohydride
Therefore, the product formed will be as follows:
(d)
Interpretation:
Draw the structural formula the compound formed by the reaction of given compound with sodium borohydride.
Concept Introduction:
Aldehydes are reduced to primary alcohols and ketones are reduced in secondary alcohols. The most commonly used reagent for reduction of aldehydes and ketone is sodium borohydride
Answer to Problem 17.65P
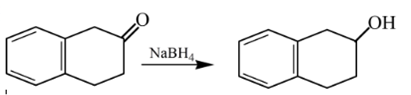
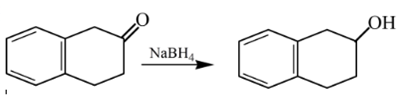
Explanation of Solution
Aldehydes are reduced to primary alcohols and ketones are reduced in secondary alcohols. The most commonly used reagent for reduction of aldehydes and ketone is sodium borohydride
Therefore, the product formed will be as follows:
(e)
Interpretation:
Draw the structural formula the compound formed by the reaction of given compound with sodium borohydride.
Concept Introduction:
Aldehydes are reduced to primary alcohols and ketones are reduced in secondary alcohols. The most commonly used reagent for reduction of aldehydes and ketone is sodium borohydride
Answer to Problem 17.65P
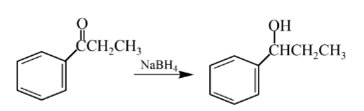
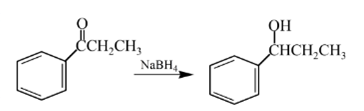
Explanation of Solution
Aldehydes are reduced to primary alcohols and ketones are reduced in secondary alcohols. The most commonly used reagent for reduction of aldehydes and ketone is sodium borohydride
Therefore, the product formed will be as follows:
(f)
Interpretation:
Draw the structural formula the compound formed by the reaction of given compound with sodium borohydride.
Concept Introduction:
Aldehydes are reduced to primary alcohols and ketones are reduced in secondary alcohols. The most commonly used reagent for reduction of aldehydes and ketone is sodium borohydride
Answer to Problem 17.65P
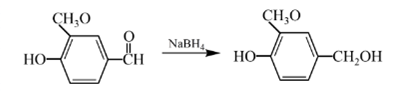
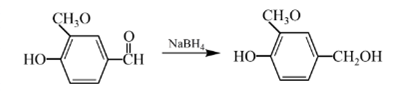
Explanation of Solution
Aldehydes are reduced to primary alcohols and ketones are reduced in secondary alcohols. The most commonly used reagent for reduction of aldehydes and ketone is sodium borohydride
Therefore, the product formed will be as follows:
Want to see more full solutions like this?
Chapter 17 Solutions
Introduction to General, Organic and Biochemistry
- 17-47 What is the characteristic structural feature of a hemiacetal? Of an acetal?arrow_forward17-29 Why can’t two molecules of acetone form a hydrogen bond with each other?arrow_forward17-33 What simple chemical test could you use to distinguish between the members of each pair of com pounds? Tell what you would do, what you would expect to observe, and how you would interpret your experimental observation. (a) Pentanal and 2-pentanone (b) 2-Pentanone and 2-pentanolarrow_forward
- 17-70 What simple chemical test could you use to distinguish between the members of each pair of com pounds? Tell what you would do, what you would expect to observe, and how you would interpret your experimental observation. (a) Benzaldehyde and cyclohexanone (b) Acetaldehyde and acetonearrow_forward17-36 Explain why the reduction of an aldehyde always gives a primary alcohol and the reduction of a ketone always gives a secondary alcohol.arrow_forward17-72 The following molecule is an enediol; each carbon of the double bond carries an —OH group. Draw structural formulas for the hydroxyketone and the a-hydroxyaldehyde with which this enediol is in equilibrium.arrow_forward
- 17-28 Show how acetaldehyde can form hydrogen bonds with water.arrow_forward17-26 Account for the fact that acetone has a higher boiling point (56°C) than ethyl methyl ether (11°C) even though their molecular weights are almost the same.arrow_forward17-35 Suppose that you take a bottle of benzaldehyde (a liquid, bp 179°C) from a shelf and find a white solid in the bottom of the bottle. The solid turns litmus red; that is, it is acidic. Yet aldehydes are neutral compounds. How can you explain these observations?arrow_forward
- 16-28 Following is the structural formula of metformin, the hydrochloride salt of which is marketed as the antidiabetic medication Glucophage. Metformin was introduced into clinical practice in the United States in 1995 for the treatment of type 2 diabetes. More than 25 million prescriptions for this drug were written in 2000, making it the most commonly prescribed brand-name diabetes medication in the nation. NH NH H3(\ 3 N N Nh2ch3 h Metformin Complete the Lewis structure for metformin, showing all valence electrons. Which nitrogen is the most likely site of protonation? Draw the structural formula of Glucophage.arrow_forward17-15 Draw structural formulas for the four aldehydes with the molecular formula C5H10O. Which of these aldehydes are chiral?arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
