
Concept explainers
A galvanic cell is based on the following half-reactions at 25°C:
Predict whether  is larger or smaller than
is larger or smaller than 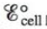 for the following cases.
for the following cases.
a.
b.
Trending nowThis is a popular solution!

Chapter 17 Solutions
Chemistry: An Atoms First Approach
Additional Science Textbook Solutions
Chemistry: The Central Science (13th Edition)
Introductory Chemistry (6th Edition)
Chemistry
Chemistry For Changing Times (14th Edition)
Chemistry: A Molecular Approach (4th Edition)
Living By Chemistry: First Edition Textbook
- An electrolysis experiment is performed to determine the value of the Faraday constant (number of coulombs per mole of electrons). In this experiment, 28.8 g of gold is plated out from a AuCN solution by running an electrolytic cell for two hours with a current of 2.00 A. What is the experimental value obtained for the Faraday Constant?arrow_forwardYou want to set up a series of voltaic cells with specific cell potentials. A Zn2+(aq, 1.0 M)| Zn(s) half-cell is in one compartment. Identify several half-cells that you could use so that the cell potential will be close to (a) 1.1 V and (b) 0.50 V. Consider cells in which the zinc cell can be either the cathode or the anode.arrow_forwardConsider a galvanic cell based on the following half-reactions: a. What is the expected cell potential with all components in their standard states? b. What is the oxidizing agent in the overall cell reaction? c. What substances make up the anode compartment? d. In the standard cell, in which direction do the electrons flow? e. How many electrons are transferred per unit of cell reaction? f. If this cell is set up at 25C with [Fe2+] = 2.00 104 M and [La3+] = 3.00 103 M, what is the expected cell potential?arrow_forward
- The half cells Sn2+(aq) |Sn(s) and Cl2(g) |Cl(aq) are linked to create a voltaic cell. (a) Write equations for the oxidation and reduction half-reactions and for the overall (cell) reaction. (b) Which half-reaction occurs in the anode compartment, and which occurs in the cathode compartment? (c) Complete the following sentences: Electrons in the external circuit flow from the ______ electrode to the ____ electrode. Negative ions move in the salt bridge from the _____ half-cell to the _____ half-cell.arrow_forwardYou have 1.0 M solutions of Al(NO3)3 and AgNO3 along with Al and Ag electrodes to construct a voltaic cell. The salt bridge contains a saturated solution of KCl. Complete the picture associated with this problem by a writing the symbols of the elements and ions in the appropriate areas (both solutions and electrodes). b identifying the anode and cathode. c indicating the direction of electron flow through the external circuit. d indicating the cell potential (assume standard conditions, with no current flowing). e writing the appropriate half-reaction under each of the containers. f indicating the direction of ion flow in the salt bridge. g identifying the species undergoing oxidation and reduction. h writing the balanced overall reaction for the cell.arrow_forwardIn principle, a battery could be made from aluminum metal and chlorine gas. (a) Write a balanced equation for the reaction thatwould occur in a battery using Al3+(aq) | Al(s) andCl2(g) | Cl(aq) half-cells. (b) Identify the half-reaction at the anode and at the cathode. Do electrons flow from the Al electrode when thecell does work? Explain. (c) Calculate the standard potential, Ecell, for the battery.arrow_forward
- Given this reaction, its standard potential, and the standard half-cell potential of 0.34 V for the Cu2+ |Cu half-cell, calculate E° for the Fe(s)|Fe2+(aq) half-cell.arrow_forwardA voltaic cell is constructed in which one half-cell consists of a silver wire in an aqueous solution of AgNO3.The other half cell consists of an inert platinum wire in an aqueous solution containing Fe2+(aq) and Fe3+(aq). (a) Calculate the cell potential, assuming standard conditions. (b) Write the net ionic equation for the reaction occurring in the cell. (c) Which electrode is the anode and which is the cathode? (d) If [Ag+] is 0.10 M, and [Fe2+] and [Fe3+] are both 1.0 M, what is the cell potential? Is the net cell reaction still that used in part (a)? If not, what is the net reaction under the new conditions?arrow_forwardA voltaic cell is constructed using the reaction of chromium metal and iron(II) ions. 2 Cr(s) + 3 Fe2+(aq) 2 Cr3+(aq) + 3 Fe(s) Complete the following sentences: Electrons in the external circuit flow from the ________ electrode to the ______ electrode. Negative ions move in the salt bridge from the ________ half-cell to the ______ half-cell. The half-reaction at the anode is _______ and that at the cathode is ________.arrow_forward
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





