
Concept explainers
The overall reaction in the lead storage battery is
Calculate 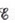 at 25°C for this battery when [H2SO4] = 4.5 M, that is, [H+] = [HSO4−] = 4.5 M. At 25°C,
at 25°C for this battery when [H2SO4] = 4.5 M, that is, [H+] = [HSO4−] = 4.5 M. At 25°C, 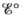 =2.04 V for the lead storage battery.
=2.04 V for the lead storage battery.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Chemistry: An Atoms First Approach
Additional Science Textbook Solutions
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Organic Chemistry
Chemistry (7th Edition)
- What is the voltage of a concentration cell of Fe2+ ions where the concentrations are 0.0025 and 0.750 M? What is the spontaneous reaction?arrow_forwardAt 298 K, the solubility product constant for PbC2O4 is 8.5 1010, and the standard reduction potential of the Pb2+(aq) to Pb(s) is 0.126 V. (a) Find the standard potential of the half-reaction PbC2O4(s)+2ePb(s)+C2O42(aq) (Hint: The desired half-reaction is the sum of the equations for the solubility product and the reduction of Pb2+. Find G for these two reactions and add them to find G for their sum. Convert the G to the potential of the desired half-reaction.) (b) Calculate the potential of the Pb/PbC2O4 electrode in a 0.025 M solution of Na2C2O4.arrow_forwardIn principle, a battery could be made from aluminum metal and chlorine gas. (a) Write a balanced equation for the reaction thatwould occur in a battery using Al3+(aq) | Al(s) andCl2(g) | Cl(aq) half-cells. (b) Identify the half-reaction at the anode and at the cathode. Do electrons flow from the Al electrode when thecell does work? Explain. (c) Calculate the standard potential, Ecell, for the battery.arrow_forward
- Another type of battery is the alkaline zinc-mercury cell, in which the cell reaction is Zn(s) + HgO(s) Hg() + ZnO(s) E = + 1.35 V (a) What is the standard free energy change for this reaction? (b) The standard free energy change in a voltaic cell is the maximum electrical energy that the cell can produce. If the reaction in a zinc-mercury cell consumes 1.00 g mercury oxide, what is the standard free energy change? (c) For how many hours could a mercury cell produce a 10-mA current if the limiting reactant is 3.50 g mercury oxide?arrow_forwardCalculate the standard cell potential of the cell corresponding to the oxidation of oxalic acid, H2C2O4, by permanganate ion. MnO4. 5H2C2O4(aq)+2MnO4(aq)+6H+(aq)10CO2(g)+2Mn2+(aq)+8H2O(l) See Appendix C for free energies of formation: Gf for H2C2O4(aq) is 698 kJ.arrow_forwardAt 298 K, the solubility product constant for Pb(IO3)2 is 2.6 1013, and the standard reduction potential of the Pb2+(aq) to Pb(s) is 0.126 V. (a) Find the standard potential of the half-reaction Pb(IO3)2(s)+2ePb(s)+2IO3(aq) (Hint: The desired half-reaction is the sum of the equations for the solubility product and the reduction of Pb2+. Find G for these two reactions, and add them to find G for their sum. Convert the G to the potential of the desired half-reaction.) (b) Calculate the potential of the Pb/Pb(IO3)2 electrode in a 3.5 103 M solution of NaIO3.arrow_forward
- Consider a voltaic cell with the reaction H2(g) + Sn4+(aq) 2 H+(aq) + Sn2+(aq) operating at 298 K. (a) Calculate the Ecell for this cell. (b) Calculate the Ecell for PH2 = 1.0 bar, (cone. Sn2+) = 6.0 104M, (cone. Sn4+) = 5.0 104M, and pH = 3.60.arrow_forwardFor each reaction listed, determine its standard cell potential at 25 C and whether the reaction is spontaneous at standard conditions. (a) Mn(s)+Ni2+(aq)Mn2+(aq)+Ni(s) (b) 3Cu2+(aq)+2Al(s)2Al3+(aq)+3Cu(s) (c) Na(s)+LiNO3(aq)NaNO3(aq)+Li(s) (d) Ca(NO3)2(aq)+Ba(s)Ba(NO3)2(aq)+Ca(s)arrow_forwardFour voltaic cells are set up. In each, one half-cell contains a standard hydrogen electrode. The second half-cell is one of the following: (i) Cr3+(aq, 1.0 M)|Cr(s) (ii) Fea+(aq, 1.0M)|Fe(s) (iii) Cu2+(aq, 1.0M)|Cu(s) (iv) Mg2+(aq, 1.0M)|Mg(s) (a) In which of the voltaic cells does the hydrogen electrode serve as the cathode? (b) Which voltaic cell produces the highest potential? Which produces the lowest potential?arrow_forward
- Use the following half-equations to write three spontaneous reactions. Justify your answers by calculating E° for the cells. 1. MnO4(aq)+8H+(aq)+5eMn2+(aq)+4H2OE=+1.512V 2. O2(g)+4H+(aq)+4e2H2OE=+1.229V 3. Co2+(aq)+2eCo(s)E=0.282Varrow_forwardFor each of the reactions, calculate E from the table of standard potentials, and state whether the reaction is spontaneous as written or spontaneous in the reverse direction under standard conditions. (a) Zn(s)+Fe2+(aq)Zn2+(aq)+Fe(s) (b) AgCl(s)+Fe2+(aq)Ag(s)+Fe3+(aq)+Cl(aq) (c) Br2(l)+2Cl(aq)Cl2(g)+2Br(aq)arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax





