
Concept explainers
a)
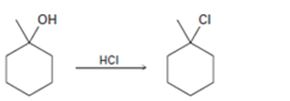
Interpretation:
The complete mechanism for the conversion of 30 alcohol given to 30
Concept introduction:
Alcohols get protonated in the presence of strong acids. The protonated intermediate will lose a molecule of water to yield a carbocation. The nucleophilic attack by the halide ion will lead to the formation of the alkyl halide.
To propose:
The complete mechanism for the conversion of 30 alcohol given to 30 alkyl halide by treating with HCl involving two cationic intermediates.
b)
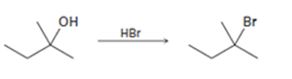
Interpretation:
The complete mechanism for the conversion of 30 alcohol given to 30 alkyl halide by treating with HBr involving two cationic intermediates is to be proposed.
Concept introduction:
Alcohols get protonated in the presence of strong acids. The protonated intermediate will lose a molecule of water to yield a carbocation. The nucleophilic attack by the halide ion will lead to the formation of the alkyl halide.
To propose:
The complete mechanism for the conversion of 30 alcohol given to 30 alkyl halide by treating with HBr involving two cationic intermediates.
c)
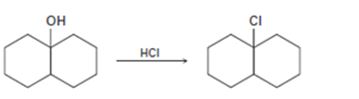
Interpretation:
The complete mechanism for the conversion of 30 alcohol given to 30 alkyl halide by treating with HCl involving two cationic intermediates is to be proposed.
Concept introduction:
Alcohols get protonated in the presence of strong acids. The protonated intermediate will lose a molecule of water to yield a carbocation. The nucleophilic attack by the halide ion will lead to the formation of the alkyl halide.
To propose:
The complete mechanism for the conversion of 30 alcohol given to 30 alkyl halide by treating with HCl involving two cationic intermediates.
Trending nowThis is a popular solution!

Chapter 17 Solutions
Organic Chemistry
- The conversion of 3 alcohols into alkenes under acidic conditions involves two cationic intermediates. For each reaction, draw the complete mechanism using curved arrows.arrow_forwardDraw a mechanism for the reaction below.arrow_forwardDraw a mechanism for the following reaction. Thank you!arrow_forward
- 1-Draw the mechanism of 1-methylcyclohexene with hydrogen bromide please. 2-Draw the reaction mechanism METHYL MAGNESIUM BROMIDE with PROPANAL and PROPANONE please.arrow_forwardUsing curved-arrows, propose a mechanism for the following transformation and draw the major product(s) (with clear stereochemistry when appropriate).arrow_forwarda. Draw the mechanism for the following reaction if it involves specific-acid catalysis.b. Draw the mechanism if it involves general-acid catalysis.arrow_forward
- Explain why pentane-2,4-dione forms two different alkylation products (Aor B) when the number of equivalents of base is increased from one totwo.arrow_forwardDraw a mechanism for the following reaction and explain each steparrow_forward#6r Daw a stepwise mechanism for the following reaction:arrow_forward
- Draw the products formed when A or B is treated with each reagent. In some cases, no reaction occurs. [1] C6H5Li (excess); [2] H2Oarrow_forwardAn allylic alcohol contains an OH group on a carbon atom adjacent to a C − C double bond. Treatment of allylic alcohol A with HCl forms a mixture of two allylic chlorides, B and C. Draw a stepwise mechanism that illustrates how both products are formed.arrow_forwardAn allylic alcohol contains an OH group on a carbon atom adjacent to a C—C double bond. Treatment of allylic alcohol A with HCl forms a mixture of two allylic chlorides, B and C. Draw a stepwise mechanism that illustrates how both products are formed.arrow_forward
