
a)
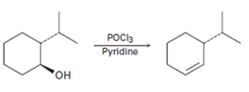
Interpretation:
A mechanism using curved arrows, for the conversion of alcohol given into the corresponding alkene by treating with POCl3, is to be proposed. The regiochemistry of the elimination is also to be explained.
Concept introduction:
The dehydration of alcohols to
To propose:
A mechanism using curved arrows, for the conversion of alcohol given into the corresponding alkene by treating with POCl3 is to be proposed.
To explain:
The regiochemistry of the elimination reaction.
b)
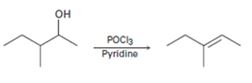
Interpretation:
A mechanism using curved arrows, for the conversion of alcohol given into the corresponding alkene by treating with POCl3, is to be proposed. The regiochemistry of the elimination is also to be explained.
Concept introduction:
The dehydration of alcohols to alkenes in the presence of POCl3 follows E2 mechanism. The –OH group is converted into a better leaving group dichlorophosphate, –OPOCl2, when treated with POCl3. Pyridine used as solvent is also a base and it removes a proton arranged anti to the leaving group from the β carbon (requirement for E2 mechanism). The removal of the proton and –OPOCl2 occurs simultaneously in a single step to yield the alkene as the product.
To propose:
A mechanism using curved arrows, for the conversion of alcohol given into the corresponding alkene by treating with POCl3, is to be proposed.
To explain:
The regiochemistry of the elimination reaction.
c)
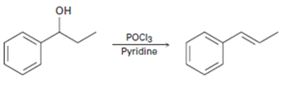
Interpretation:
A mechanism using curved arrows, for the conversion of alcohol given into the corresponding alkene by treating with POCl3, is to be proposed. The regiochemistry of the elimination is also to be explained.
Concept introduction:
The dehydration of alcohols to alkenes in the presence of POCl3 follows E2 mechanism. The –OH group is converted into a better leaving group dichlorophosphate, –OPOCl2, when treated with POCl3. Pyridine used as solvent is also a base and it removes a proton arranged anti to the leaving group from the β carbon (requirement for E2 mechanism). The removal of the proton and –OPOCl2 occurs simultaneously in a single step to yield the alkene as the product.
To propose:
A mechanism using curved arrows, for the conversion of alcohol given into the corresponding alkene by treating with POCl3, is to be proposed.
To explain:
The regiochemistry of the elimination reaction.
Trending nowThis is a popular solution!

Chapter 17 Solutions
Organic Chemistry
- 1. (1-bromo-1, 3-dimethylcyclopentane) an optically active pure sample reacted with water. Write the complete mechanism which includes the electron-pushing arrows. Show the expected stereochemistry for each of the case and each mechanism should give two distinct products and then describe the relationship of the products to one another.arrow_forwardDraw the major product formed when HBr reacts with the following epoxide. Use wedge/dash bonds, including H's at each stereogenic center, to show the stereochemistry of the product.arrow_forwardIgnoring stereoisomers, draw the two possible enols for butan-2-one (CH3COCH2CH3), and predict which one is more stable.arrow_forward
- Based on the more stable conformation (conformation A), need help providing two mechanisms for the E2 elimination of the protonated version of cis-1 using a general base (B:) to give alkenes 2 and 3. Clearly show which hydrogens meet the stereochemical requirements for E2-elimination Using Zaitsev’s rule, indicate which is favoured. Pictured is also a reaction mechanism to show what the alkenes 2 and 3 are. Thank you :)arrow_forwardOxidation of cholesterol converts the alcohol to a ketone. Under acidic or basic oxidationconditions, the C“C double bond migrates to the more stable, conjugated position. BeforeIR and NMR spectroscopy, chemists watched the UV spectrum of the reaction mixture tofollow the oxidation. Describe how the UV spectrum of the conjugated product, cholest-4-en-3-one, differs from that of cholesterolarrow_forwardwhat are the products? please indicate the relative stereochemistry for the following transformations and indicate the major product.arrow_forward
- Draw the products formed when both cis- and trans-but-2-ene are treated with a peroxyacid followed by -OH (in H2O).Explain how these reactions illustrate that anti dihydroxylation is stereospecic.arrow_forward(a) When (Z)-3-methylhex-3-ene undergoes hydroboration–oxidation, two isomericproducts are formed. Give their structures, and label each asymmetric carbon atomas (R) or (S). What is the relationship between these isomers?(b) Repeat part (a) for (E)-3-methylhex-3-ene. What is the relationship between the productsformed from (Z)-3-methylhex-3-ene and those formed from (E)-3-methylhex-3-ene?arrow_forwardConsider a reaction where cis-but-2-ene is treated with a peroxy acid followed by OH- /H20. Draw the structure of one product that is formed in the reaction, including correct stereochemistry.arrow_forward
- Using cis- and trans-hex-3-ene, demonstrate that the addition of HCl is not a stereospecific reaction. Draw the structure of the stereoisomers formed from each alkene.arrow_forwardDraw the products formed when each alkene is treated with BH3 followed by H2O2,HO−. Include the stereochemistry at all stereogenic centers.arrow_forwardDraw the major product formed when HBrHBr reacts with the epoxide. Use wedge–dash bonds, including hydrogen atoms at each stereogenic center, to show the stereochemistry of the product.arrow_forward
