
(a)
Interpretation:
The structural formula for the given compound has to be drawn.
Concept introduction:
Nomenclature of esters of carboxylic acids:
The acyl group bonded to
The naming of an ester of a
(a)
Explanation of Solution
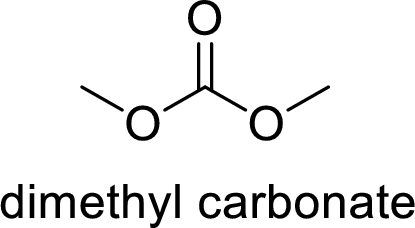
The name of the compound is given below:
Dimethyl carbonate
From the name, it is clear that two methyl groups are attached to oxygen atoms in the compound and the parent ester group is carbonate.
Thus, the structure of the compound is given below:
(b)
Interpretation:
The structural formula for the given compound has to be drawn.
Concept introduction:
Nomenclature of nitriles:
The numbering 1 can be given to the nitrile carbon atom and add nitrile to end of the
(b)
Explanation of Solution
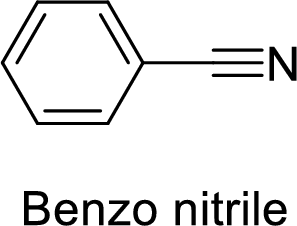
The name of the compound is given below:
The functional group is nitrile and benzene ring is attached to it.
Thus, the structure of the compound is given below:
Benzo nitrile
(c)
Interpretation:
The structural formula for the given compound has to be drawn.
Concept introduction:
Nomenclature of esters of carboxylic acids:
The acyl group bonded to
The naming of an ester of a carboxylic acid is done by naming alkyl or aryl group bonded to oxygen first followed by the name of the acid in which the suffix –ic acid is replaced by the suffix –ate.
(c)
Explanation of Solution
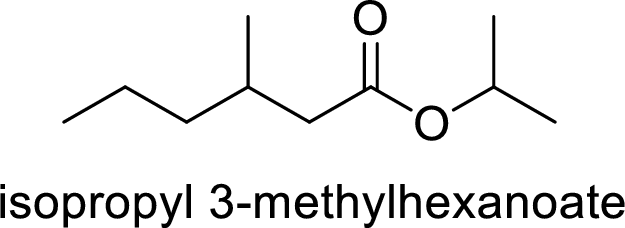
The name of the compound is given below:
Isopropyl-3-methylhexanoate
The alkyl group attached on oxygen atom is the isopropyl group and the parent chain is the methylhexanoate.
Thus, the structure of the compound is given below:
(d)
Interpretation:
The structural formula for the given compound has to be drawn.
Concept introduction:
Nomenclature of esters of carboxylic acids:
The acyl group bonded to
The naming of an ester of a carboxylic acid is done by naming alkyl or aryl group bonded to oxygen first followed by the name of the acid in which the suffix –ic acid is replaced by the suffix –ate.
(d)
Explanation of Solution
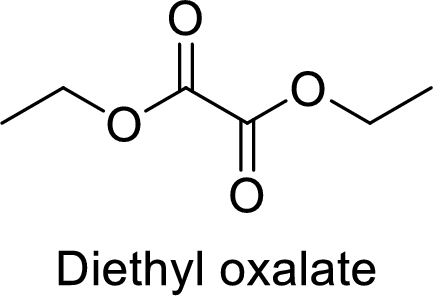
The name of the compound is given below:
Diethyl oxalate
From the name, it is clear that two ethyl groups are attached to oxygen atoms in the compound and the parent ester group is oxalate.
Thus, the structure of the compound is given below:
(e)
The structural formula for the given compound has to be drawn.
Concept introduction:
Nomenclature of esters of carboxylic acids:
The acyl group bonded to
The naming of an ester of a carboxylic acid is done by naming alkyl or aryl group bonded to oxygen first followed by the name of the acid in which the suffix –ic acid is replaced by the suffix –ate.
(e)
Explanation of Solution
The name of the compound is given below:
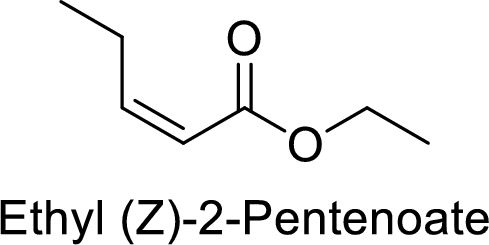
Ethyl (Z)-2-pentenoate
The alkyl group attached on oxygen atom is the ethyl group and the parent chain is the pentenoate. The double bond is having E configuration.
Thus, the structure of the compound is given below:
(f)
Interpretation:
The structural formula for the given compound has to be drawn.
Concept introduction:
Nomenclature of anhydrides:
The nomenclature of anhydride involves replacement of the word acid with anhydride. In the case of mixed anhydrides, both acids should be named.
(f)
Explanation of Solution
The name of the compound is given below:
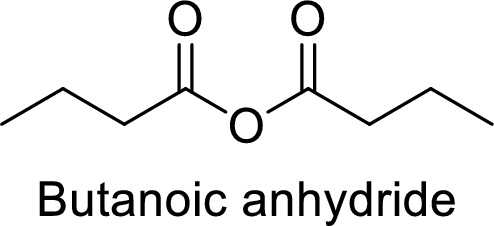
Butanoic anhydride
From the butanoic acid, acid part can be replaced with an anhydride group.
(g)
Interpretation:
The structural formula for the given compound has to be drawn.
Concept introduction:
Nomenclature of amides and imides:
The acyl group bonded to a nitrogen atom is the functional group of an amide.
The naming of an amide can be done by giving the suffix –oic acid from the IUPAC name of the parent acid and adding –amide.
The location of the alkyl or aryl group attached on nitrogen atom in an amide is indicated by ‘N-‘
The indication “ N,N-di-“ is used for two identical alkyl or aryl groups on nitrogen atom
(g)
Explanation of Solution
The name of the compound is given below:
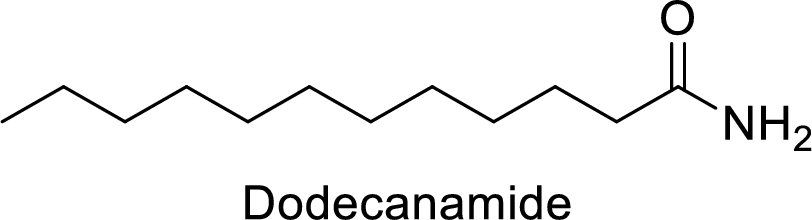
Dodecanamide:
The functional group is amide and the acyl group attached is the dodecane group.
Thus, the structure of the compound is given below:
(h)
The structural formula for the given compound has to be drawn.
Concept introduction:
Nomenclature of esters of carboxylic acids:
The acyl group bonded to
The naming of an ester of a carboxylic acid is done by naming alkyl or aryl group bonded to oxygen first followed by the name of the acid in which the suffix –ic acid is replaced by the suffix –ate.
(h)
Explanation of Solution
The name of the compound is given below:
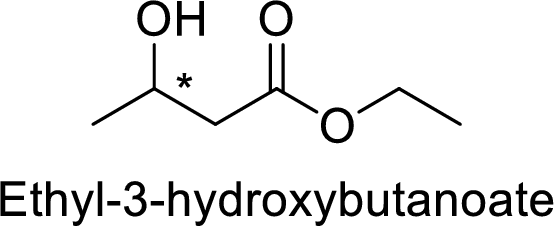
Ethyl-3-hydroxybutanoate
The alkyl group attached on oxygen atom is the ethyl group and the parent chain is the butanoate and a hydroxyl group is present in the third position.
Thus, the structure of the compound is given below:
(i)
The structural formula for the given compound has to be drawn.
Concept introduction:
Nomenclature of acid chlorides:
The nomenclture of acid chlorides can be done by replacing –oic acid with –oyl chloride.
(i)
Explanation of Solution
The name of the compound is given below:
Octanoyl chloride
The acid group from the octanoic acid will be replaced by acid chloride group.
Thus, the structure of the compound is given below:
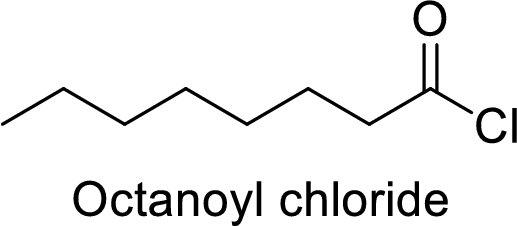
(j)
The structural formula for the given compound has to be drawn.
Concept introduction:
Nomenclature of esters of carboxylic acids:
The acyl group bonded to
The naming of an ester of a carboxylic acid is done by naming alkyl or aryl group bonded to oxygen first followed by the name of the acid in which the suffix –ic acid is replaced by the suffix –ate.
(j)
Explanation of Solution
The name of the compound is given below:
Diethyl cis-1,2-cyclohexanedicarboxylate
The alkyl group attached on oxygen atom are two ethyl groups and the parent chain is the dicarboxylate and a cyclohecxane group is present in the 1,2 position.
Thus, the structure of the compound is given below:
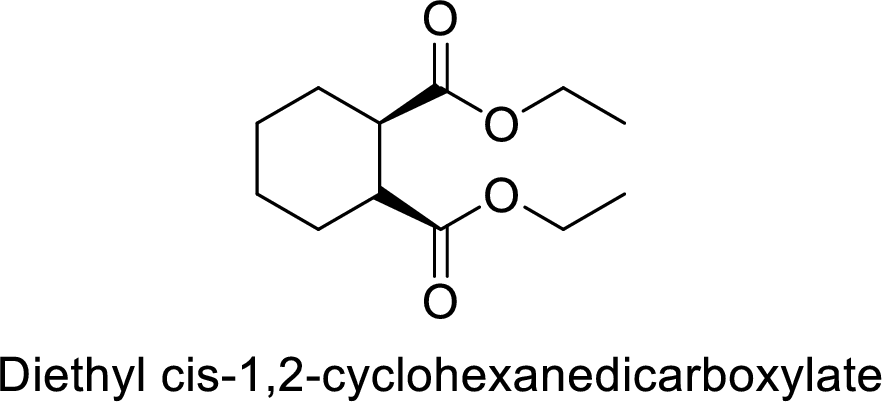
(k)
The structural formula for the given compound has to be drawn.
Concept introduction:
Nomenclature of acid chlorides:
The nomenclture of acid chlorides can be done by replacing –oic acid with –oyl chloride.
(k)
Explanation of Solution
The name of the compound is given below:
Methanesulfonyl chloride
The acid group from the methanesulfonic acid will be replaced by acid chloride group.
Thus, the structure of the compound is given below:
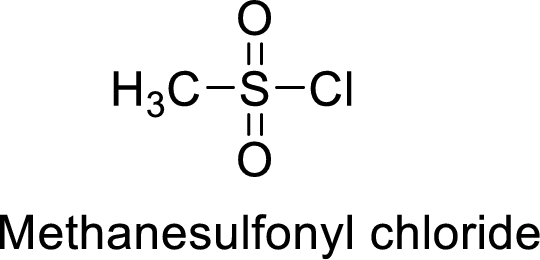
(l)
The structural formula for the given compound has to be drawn.
Concept introduction:
Nomenclature of acid chlorides:
The nomenclture of acid chlorides can be done by replacing –oic acid with –oyl chloride.
(l)
Explanation of Solution
The name of the compound is given below:
p-Toluenesulfonyl chloride
The acid group from the p-toluenesulfonic acid will be replaced by acid chloride group.
Thus, the structure of the compound is given below:
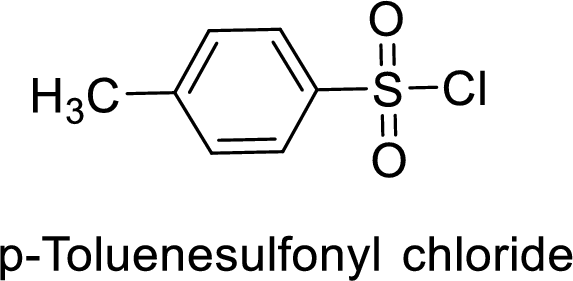
Want to see more full solutions like this?
Chapter 18 Solutions
Organic Chemistry
- (a) Compound Z is a tertiary aromatic amine with the formula, C8H11N. Provide a chemical structure for compound Z. (b)nDraw the structure of the product formed exclusively when nitrous acid reacts with Z.arrow_forwardAssume that you have samples of the following two compounds, both with formula C7H8O. Both compounds dissolve in ether, but only one of the two dissolves in aqueous NaOH. How could you use this information to distinguish between them?arrow_forwardDraw the structure of sodium tetradecyl sulfate and ciprofloxacin and state how the activity of the drug is influenced by alkane groupings.arrow_forward
- Give the expected organic product when phenylacetic acid, PhCH2COOH, is treated with reagent Q.)LiAlH4 followed by H2Oarrow_forwardGrignard reagent is a versatile tool in synthetic organic chemistry. Using bromocyclopentane as a starting material, show how a Grignard reagent, X, is synthesized. Reaction of X with water produces compound Y while treatment in carbon dioxide followed by hydrolysis forms compound Z. 3-methyl-2butanone reacts with X and hydrolyses to yield compound AA. Draw the structural formulae of compounds Y, Z and AA and write the chemical equations respectively.arrow_forwardTreatment of salicylaldehyde (2-hydroxybenzaldehyde) with bromine in glacial acetic acid at 0°C gives a compound with the molecular formula C7H4Br2O2, which is used as a topical fungicide and antibacterial agent. Propose a structural formula for this compoundarrow_forward
- Following is the structural formula of the tranquilizer meparfynol (Oblivon). Propose a synthesis for this compound starting with acetylene and a ketone. (Notice the -yn- and -ol in the chemical name of this compound, indicating that it contains alkyne and hydroxyl functional groups.)arrow_forwardDoes it safe to use this sunscreen K on acne prone skin. And can we rinse off it by only using facial gel mixed with water to rinse off it without use oil. Do I need to use micellar water for removing cosmetic before rinse off it with facial gel Sunscreen K has lists of ingredients below (descending order)…. AQUA DIMETHICONE CYCLOPENTASILOXANE PROPYLENE GLYCOL NIACINAMIDE DIETHYLHEXYL CARBONATE ZINC OXIDE CETEARYL ALCOHOL POLYMETHYL METHACRYLATE SODIUM POLYACRYLOYL-DIMETHYL TAURATE TITANIUM DIOXIDE ETHYLHEXYL METHOXYCINNAMATE BUTYL METHOXYDIBENZOYLMETHANE GLYCERYL STEARATE BIS-PEG/PPG-20/5 PEG/PPG-20/5 DIMETHICONE METHOXY PEG/PPG-25/4 DIMETHICONE HYDROGENATED POLYDECENE BUTYLENE GLYCOL OCTOCRYLENE PEG-40 STEARATE PHENOXYETHANOL HEXYL LAURATE POLYHYDROXYSTEARIC ACID CAPRYLIC/CAPRIC TRIGLYCERIDE STEARIC ACID CETYL ALCOHOL PEG-10 DIMETHICONE CETEARETH-20 TOCOPHERYL ACETATE STEARYL ALCOHOl PHENYL TRIMETHICONE TRIETHOXYCAPRYLYSILANE TRIDECETH-10 CHLORPHENESIN DISODIUM EDTA DIMETHICONOL…arrow_forwardThe oxidation of 3-methylbutanal with potassium permanganate in an acid medium produces compound A and the reduction of cyclohexanone with NaBH4 produces compound B. Indicate the CORRECT alternative: a) A reacts rapidly with water to produce a carboxylic acid. b) Reaction of B with methylamine produces an amide. c) Reaction of A with an acyl chloride produces an ester. d) B reacts with HCN to produce a cyanohydrin. e) The reaction of A and B in an acid medium produces cyclohexyl 3-methylbutanoate.arrow_forward
- Benzoic acid, Ph-COOH (C6H5CO2H), is not soluble in water while it dissolves in ether (diethyl ether), (CH3CH2)2O. Yet upon treatment with sodium hydroxide, benzoic acid turns hydrophilic and dissolves in water. Provide chemical explanation of this observation.arrow_forwardGive the expected organic product when phenylacetic acid, PhCH2COOH, is treated with reagent Q.)NaOH, H2Oarrow_forwardWhen acetophenone, hydroxelamine hydrochloride, and sodium hydroxide are reacted to synthesize acetophenone oxime, why is it necessary to add hydrochloric acid after the reaction to make it acidic?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
