
(a)
Interpretation:
The increasing order of
Concept introduction:
The nucleophilic substitution reactions are the reactions in which one nucleophile is substituted by another nucleophile. These reactions depend upon the nucleophilicity and concentration of the nucleophile. It is of two types,
The
The
Answer to Problem 18.1P
The given compounds with the increasing order of
Explanation of Solution
The structure of given compounds is shown below
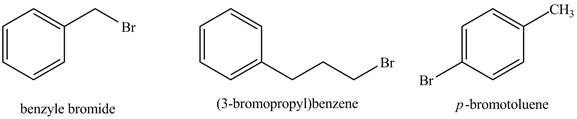
Figure 1
The
In
Therefore, the given compounds with the increasing order of
The increasing order of
(b)
Interpretation:
The increasing order of
Concept introduction:
The nucleophilic substitution reactions are the reactions in which one nucleophile is substituted by another nucleophile. These reactions depend upon the nucleophilicity and concentration of the nucleophile. It is of two types,
The
The
Answer to Problem 18.1P
The given compounds with the increasing order of
Explanation of Solution
The structure of given compounds is shown below.
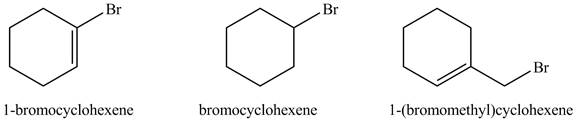
Figure 2
The
In
The increasing order of
Want to see more full solutions like this?
Chapter 18 Solutions
Organic Chemistry
- Does the reaction of S-2-bromobutane with sodium hydroxide produce a racemic mixturearrow_forwardmost of the additions of bromine to double bonds gave entirely antistereochemistry. Explain why the addition to phenanthrene gives a mixture of synand anti stereochemistry. When the product from (c) is heated, HBr is evolved and 9-bromophenanthrene results.Propose a mechanism for this dehydrohalogenation.arrow_forwardWhen 5-bromo-1-pentanol is treated with sodium hydride in diethyl ether, the product is analyzed to be C5H10O. Propose a likely structure for this product, suggesting a reasonable mechanistic pathway for its formationarrow_forward
- Rank the following compounds in decreasing order of their reactivity toward the SN2 reaction with sodium ethoxide (Na+ -OCH2CH3) in ethanol.methyl chloride tert-butyl iodide neopentyl bromide isopropyl bromide methyl iodide ethyl chloridearrow_forwardWhen 1 mole of buta-1,3-diene reacts with 1 mole of HBr, both 3-bromobut-1-ene and 1-bromobut-2-ene are formed. Propose a mechanism to account for this mixture of products.arrow_forwardIn a strongly acidic solution, cyclohexa-1,4-diene tautomerizes to cyclohexa-1,3-diene.Propose a mechanism for this rearrangement, and explain why it is energetically favorablearrow_forward
- 2-Methyl-2-butene reacts with HBr in the presence of peroxide to give a. a secondary alkyl bromide. b. a primary alkyl bromide. c. a tertiary alkyl bromide. d. a vicinal dibromide.arrow_forwardWhen 2,2-dibromo-1-phenylpropane is heated overnight in fused KOH at 200 °C, the major product is a foul-smelling compound of formula C9H8. Propose a structure for this product, and give a mechanism to account for its formation.arrow_forwardodine monochloride, ICI, is a reagent whose reactivity mirrors that of other dihalogens (X₂) with nucleophilic πt bonds. Using curved arrows to show the flow of electrons, draw a plausible mechanism for the following transformation. Make sure that your mechanism accounts for the correct stereoselectivity.arrow_forward
- Provide the mechanism for the following transformation. D = 2Harrow_forwardThe reaction of butan-2-ol with concentrated aqueous HBr goes with partial racemization, giving more inversion thanretention of configuration. Propose a mechanism that accounts for racemization with excess inversion.(b) Under the same conditions, an optically active sample of trans-2-bromocyclopentanol reacts with concentrated aqueous HBr to give an optically inactive product, (racemic) trans-1,2-dibromocyclopentane. Proposea mechanism to show how this reaction goes with apparently complete retention of configuration, yet withracemization. (Hint: Draw out the mechanism of the reaction of cyclopentene with Br2 in water to give thestarting material, trans-2- bromocyclopentanol. Consider how parts of this mechanism might be involved in thereaction with HBr.)arrow_forward8a)Provide mechanisms for the following reactions:arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
