
Interpretation:
The product of the reaction of propanal with the given reagents has to be stated.
Concept Introduction:
Lithium aluminum hydride and sodium borohydride are strong reducing agents. They are inorganic compounds which are used as the reducing agents in
In the reaction of
By catalytic hydrogenation, aldehydes are reduced to primary alcohols. Grignard reagents react with ketones and aldehydes to form alcohols. These reactions are nucleophilic addition reactions. The Grignard reagent adds to the carbonyl group of aldehydes and ketones due to electronegativity difference between carbon and oxygen.
An organolithium reagent acts like a good nucleophiles and strong bases. They used for the conversion of aldehydes and ketones into primary and secondary alcohols. Acetal is an organic compound with general formula
Answer to Problem 27P
Solution:
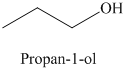
a) The product of the reaction of propanal with lithium aluminum hydride, followed by water is shown below.
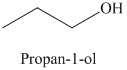
b) The product of the reaction of propanal with sodium borohydride, methanol is shown below.
c) The product of the reaction of propanal with hydrogen (nickel catalyst) is shown below.
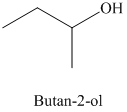
d) The product of the reaction of propanal with methylmagnesium iodide, followed by dilute acid is shown below.
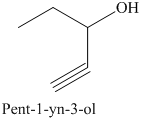
e) The product of the reaction of propanal with sodium acetylide, followed by dilute acid is shown below.
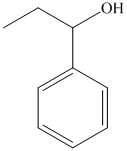
f) The product of the reaction of propanal with phenyllithium, followed by dilute acid is shown below.
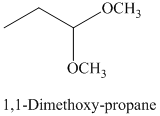
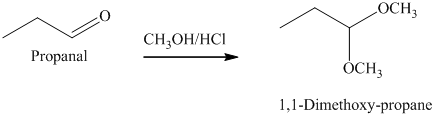
g) The product of the reaction of propanal with methanol containing dissolved hydrogen chloride is shown below.
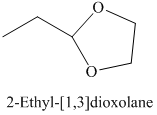
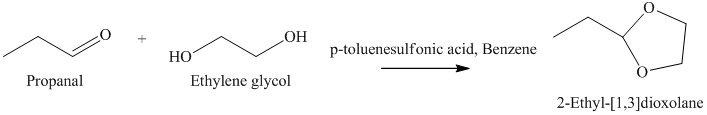
h) The product of the reaction of propanal with ethylene glycol, p-toluenesulfonic acid, benzene is shown below.
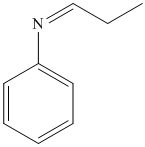
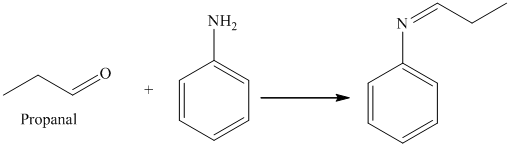
i) The product of the reaction of propanal with aniline
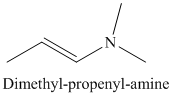
j) The product of the reaction of propanal with dimethylamine, p-toluenesulfonic acid, benzene is shown below.
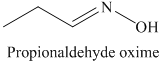
k) The product of the reaction of propanal with hydroxylamine is shown below.
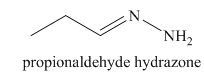
l) The product of the reaction of propanal with hydrazine is shown below.
m) The product of the reaction of propanal with product of part (l) heated in triethylene glycol with sodium hydroxide is shown below.
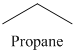
n) The product of the reaction of propanal with p-Nitrophenylhydrazine is shown below.
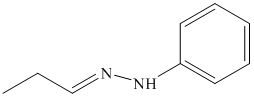
o) The product of the reaction of propanal with semicarbazide is shown below.
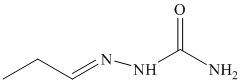
p) The product of the reaction of propanal with ethylidenetriphenylphosphorane is shown below.
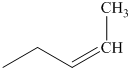
q) The product of the reaction of propanal with sodium cyanide with addition of sulfuric acid is shown below.
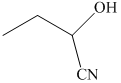
r) The product of the reaction of propanal with chromic acid is shown below.
Explanation of Solution
a) The product obtained by the reaction between, propanal and the reagent, lithium aluminum hydride, followed by water.
The reaction of propanal with lithium aluminum hydride, followed by water gives primary alcohol as the final product. The product of this reaction is shown below.
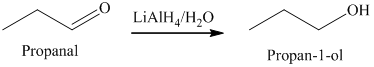
b) The product obtained by the reaction between propanal and the reagent, sodium borohydride, methanol.
The reaction of propanal with sodium borohydride, followed by methanol gives primary alcohol as the final product. The product of this reaction is shown below.
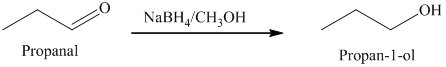
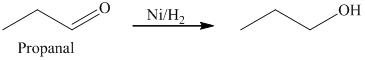
c) The product obtained by the reaction between the given compound, propanal and the reagent, hydrogen (nickel catalyst).
The reaction of propanal with hydrogen in the presence of nickel catalyst gives
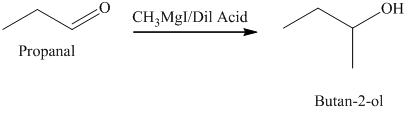
d) The product obtained by the reaction between propanal and the reagent, methylmagnesium iodide, followed by dilute acid.
The reaction of propanal with methylmagnesium iodide that is Grignard reagent, followed by dilute acid gives alcohol as the final product. The product of this reaction is shown below.
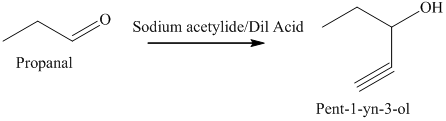
e) The product obtained by the reaction between propanal and the reagent, sodium acetylide, followed by dilute acid.
The reaction of aldehyde with sodium acetylide is fundamentally similar to the Grignard reaction. The reaction of propanal with sodium acetylide, followed by dilute acid gives alcohol. The product of this reaction is shown below.
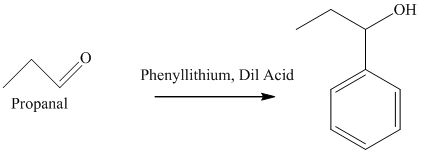
f) The product obtained by the reaction between propanal and the reagent, phenyllithium, followed by dilute acid.
The reaction of propanal with phenyllithium, followed by dilute acid gives alcohol as the final product. The product of this reaction is shown below.
g) The product obtained by the reaction between the given compound, propanal and the reagent, methanol containing dissolved hydrogen chloride.
The reaction of aldehydes with two equivalents of an alcohol results in the formation of acetals. The product of this reaction is shown below.
h) The product obtained by the reaction between the given compound, propanal and the reagent, Ethylene glycol, p-toluenesulfonic acid, benzene.
In the reaction of aldehyde with ethylene glycol, p-toluenesulfonic acid and benzene, the protection of the carbonyl group of aldehyde takes place. For carbonyl protection, ethylene glycol is the commonly used group. The final product resembles like ether and known as ketal during the protection of carbonyl group using ethylene glycol. The product of this reaction is shown below.
i) The product obtained by the reaction between propanal and the reagent, aniline
The reaction of aldehyde with primary
j) The product obtained by the reaction between propanal and the reagent, dimethylamine, p-toluenesulfonic acid, benzene.
The reaction of aldehyde with secondary amine forms enamine as the final product. The reaction of propanal with dimethylamine in the presence of p-toluenesulfonic acid and benzene gives

k) The product obtained by the reaction between the given compound, propanal and the reagent, hydroxylamine.
The reaction of aldehyde with hydroxylamine gives oxime as the final product. The reaction of propanal with hydroxylamine results in the formation of propionaldehyde oxime. The product of this reaction is shown below.
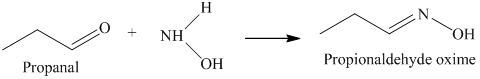
l) The product obtained by the reaction between the given compound, propanal and the reagent, hydrazine.
The reaction of aldehyde with hydrazine gives hydrazone. The reaction of propanal with hydrazine gives propionaldehyde hydrazone as the final product. The product of this reaction is shown below.
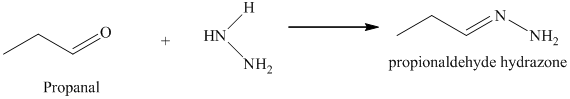
m) The product obtained by the reaction between propanal and the product of part (l) heated in triethylene glycol with sodium hydroxide.
The reaction of aldehyde with hydrazine gives hydrazone. The reaction of propanal with hydrazine gives propionaldehyde hydrazone as the final product. The heating of propionaldehyde hydrazone in triethylene glycol with sodium hydroxide forms

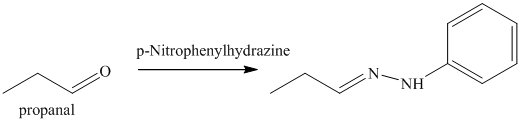
n) The product obtained by the reaction between propanal and p-nitrophenylhydrazine.
The reaction of aldehyde with hydrazine gives hydrazone. The reaction of propanal with p-Nitrophenylhydrazine gives propionaldehyde phenylhydrazone as the final product. The product of this reaction is shown below.
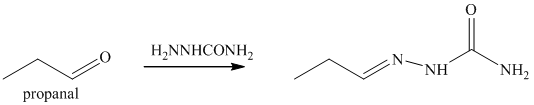
o) The product obtained by the reaction between propanal and semicarbazide.
The reaction of aldehyde with semicarbazide results in the formation of semicarbazone. The reaction of propanal with
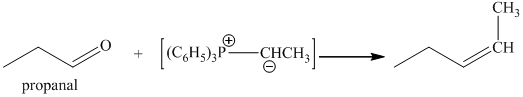
p) The product obtained by the reaction between the given compound, propanal and ethylidenetriphenylphosphorane.
The reaction of propanal with ethylidenetriphenylphosphorane gives
q) The product obtained by the reaction between propanal and sodium cyanide with addition of sulfuric acid.
The reaction of aldehyde with sodium cyanide results in the formation of cyanohydrin. The product of this reaction is shown below.
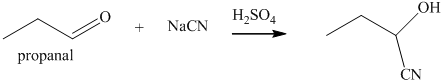
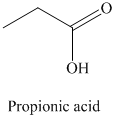
r) The product obtained by the reaction between propanal and chromic acid.
The reaction of propanal with chromic acid gives propionic acid as the final product. The product of this reaction is shown below.

Want to see more full solutions like this?
Chapter 18 Solutions
Organic Chemistry - Standalone book
- The insecticide DDT is prepared by the following route. Suggest a mechanism for this reaction. The abbreviation DDT is derived from the common name dichlorodiphenyltrichloroethane.arrow_forwardThe synthesis of triphenylphosphine was carried out. Firstly the synthesis of the reagent, phenyl magnesium bromide followed by the addition of phosphorous tribromide which was further quenched, extracted and purified to give triphenylphosphine as a product. Write a reaction mechanism for this reaction. Discuss the reaction and what occurs.arrow_forwardCompound A exhibits the following H1 NMR, 13C NMR, and partial mass spectra respectively. When compound A is hydrolyzed, compound B is produced, Compound B exhibits the following H1 NMR, 13C NMR, and partial mass spectra respectively. Suggest structures for compounds A and B.arrow_forward
- Following is a series of anorexics (appetite suppressants). As you study their structures, you will surely be struck by the sets of characteristic structural features Q. Knowing what you do about the synthesis of amines, including the Ritter reaction , suggest a synthesis for compoundarrow_forwardThe following three derivatives of succinimide are anticonvulsants that have found use in the treatment of epilepsy, particularly petit mal seizures. Q. Of these three anticonvulsants, one is considerably more acidic than the other two. Which is the most acidic compound? Estimate its pKa and account for its acidity. How does its acidity compare with that of phenol? with that of acetic acid?arrow_forwardA compound with molecular formula C8H8O3 exhibits the following IR, 1H NMR and 13C NMR spectra. Deduce the structure of this compound.arrow_forward
- Explain how benzaldehyde and dimedone reacts with each other, and then with the aminotriazole to form compound 1a in the presence of an acid catalyst. Provide a detailed reaction mechanism and explanation.arrow_forwardWhen 3-iodo-2,2-dimethylbutane is treated with silver nitrate in ethanol, three elimination products are formed. Give their structures, and predict which ones are formed inlarger amounts.arrow_forwardWhen 1,4-dibromobutane was treated with excess ethanamine at propanone, the main product had the molecular formula C6H13N. Deduce the structure of this product by proposing a mechanism for the reaction.arrow_forward
- A compound with molecular formula C5H10O2 gives the following IR spectrum. When it undergoes acid-catalyzed hydrolysis, the compound with the 1H NMR spectrum shown below is formed. Identify the compounds.arrow_forwardA synthesis of the pain reliever phenacetin is outlined in the following equation. What is the structure of phenacetin?arrow_forwardPropose the structure of a starting material that would allow you to use the procedures given to synthesize acetaminophenarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

