
Concept explainers
Interpretation:
The structural formula corresponding to the given name has to be stated.
Concept Introduction:
The systematic naming of organic compound is given by
Rules for writing structural formula from IUPAC are as follows:
First identify the word root for the given compound.
Identify the suffix used in the given compound like –ene, oic, ane, etc.
Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 13P
Solution:
a) The structural formula corresponding to the given name is shown below.
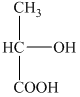
b) The structural formula corresponding to the given name is shown below.
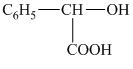
c) The structural formula corresponding to the given name is
d) The structural formula corresponding to the given name is
e) The structural formula corresponding to the given name is shown below.
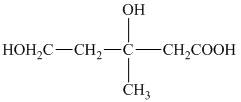
f) The structural formula corresponding to the given name is shown below.
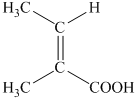
g) The structural formula corresponding to the given name is shown below.
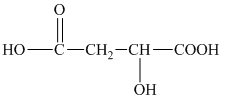
h) The structural formula corresponding to the given name is shown below.
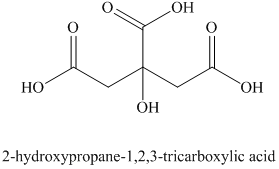
i) The structural formula corresponding to the given name is shown below.
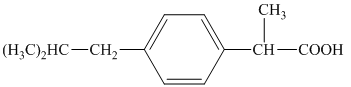
j) The structural formula corresponding to the given name is shown below.
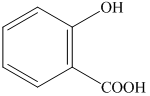
Explanation of Solution
a) Structural formula of
The given name is
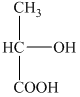
b) Structural formula of
The given name is
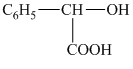
c) Structural formula of tetradecanoic acid.
The given name is tetradecanoic acid. The word root used in this is tetradec. It means structure contains fourteen carbon atoms. The functional group present in the given compound is carboxylic acid
d) Structural formula of
The given name is
e) Structural formula of
The given name is
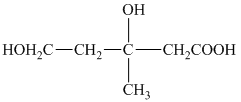
f) Structural formula of
The given name is
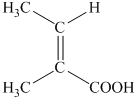
g) Structural formula of
The given name is
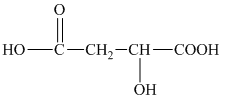
h) Structural formula of
The given name is
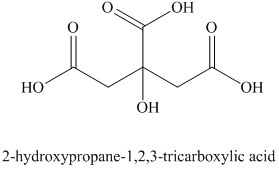
i) Structural formula of
The given name is
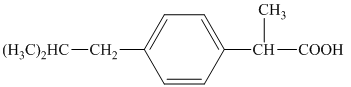
j) Structural formula of
The given name is
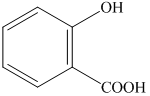
Want to see more full solutions like this?
Chapter 19 Solutions
ORGANIC CHEMISTRY TEXT PACKAGED - 2 YE
- When the conjugate acid of aniline, C6H5NH3+, reacts with the acetate ion, the following reaction takes place: C6H5NH3+(aq)+CH3COO(aq)C6H5NH2(aq)+CH3COOH(aq) If Kafor C6H5NH3+ is 1.35105 and Kafor CH3COOH is 1.86105 , what is K for the reaction?arrow_forwardWhat are 2-3 organic compounds that each contain at least one of the following: carboxylic acid, ester, amine, or amide. How it is synthesized and used, and how the function of the compound is facilitated by its structure and, specifically, its functional groups.arrow_forwardWhich of the following straight-chain organic compounds has the highest acidity? (Note: All numbers in formulas are subscripts.)A. C2H2B. C2H6C. CH4D. C2H4E. C4H10arrow_forward
- Write the chemical equation for a pentanoate ion acting as base when it reacts with hydrochloric acid (HCI). Which is formula of this rule of reaction?arrow_forwardPlease help in answering the following questions ~Which of the following is the basis of the classification of alcohols?A. Based on the number of hydroxyl groups attached.B. Based on the number of carbon atoms which are directly attached to the carbon thatis bonded with the -OH group.C. BothD. Neither ~Which of the following carboxylic acids will yield the least amount of protons in an aqueoussolution?A. 2-chlorobutanoic acidB. Propanoic acidC. Butanoic acidD. 2-methylbutanoic acidarrow_forward7. The following compounds are isomeric esters derived from acetic acid, each with formula C5H10O2. Draw the structures of the two esters?arrow_forward
- In an advanced synthetic chemistry experiment, a researcher prepares a compound, ZY-7, by reacting a ketone (C5H10O) with hydroxylamine (NH2OH), followed by heating in the presence of an acid catalyst. The resulting compound, ZY-7, is then treated with a solution of sodium nitrite (NaNO2) and hydrochloric acid (HCl) at low temperature. Identify the class of compound that ZY-7 most likely belongs to after this series of reactions." A) Amide B) Oxime C) Nitro compound D) Diazonium salt E) Ester Don't use chatgpt please provide valuable answerarrow_forwardDiazomethane, CH2N2, is used in the organic chemistry laboratory despite its danger because it produces very high yields and is selective for reaction with carboxylic acids. Write the products of the following reactions.arrow_forward1. Which of the following statements is true? I. Aldehydes and ketones both contain a hydroxyl group. II. The names for aldehydes and ketones are derived from the name of the longest carbon chain that contains the carbonyl group. III. The aldehyde and ketone with a molecular formula of C3H6O are constitutional isomers. IV. 2-Propanone is immiscible in water. A. I & II B. II & II C. I & III D. I & IV 2. Whicb of the following is the correct IUPAC name of the structure below?arrow_forward
- Draw the skeletal structure of the following compound: a. carboxylic acid with the molecular formula C4H8O2 b. an ester with molecular formula C4H802 c. a ketone with the molecular formula C4H8O2 d. an aldehyde with the molecular formula C4H8Oarrow_forwardPrepare CH3COOC2H5, ethyl acetate, using ethyl alcohol. Write the chemical reaction. The pain reliever acetaminophen is produced by reacting 4-aminophenol with acetic anhydride. Outline a synthesis of acetaminophen from 4-aminophenol including any needed inorganic reagents.arrow_forwardWrite an equation representing the neutralization of eachof the following carboxylic acids with KOH:a. 3-Chlorohexanoic acidb. Cyclohexanecarboxylic acidc. 3,4-Dimethylpentanoic acidarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning




