
Concept explainers
(a)
Interpretation:
The structure for the compound isobutryaldehyde is to be drawn.
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
Answer to Problem 19.1P
The structure of isobutrylaldehyde is shown below.
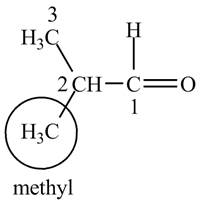
Explanation of Solution
Isobutrylaldehyde is a compound containing 4 carbon atoms and
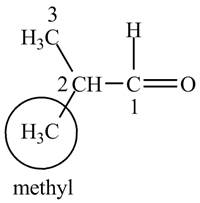
Figure 1
The IUPAC name of the compound is
The structure for the compound isobutrylaldehyde is shown in Figure 1.
(b)
Interpretation:
The structure for the compound valerophenone is to be drawn.
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
Answer to Problem 19.1P
The structure of valerophenone is given below.
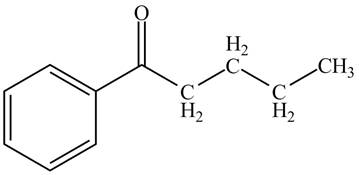
Explanation of Solution
Valerophenone is the name of the compound having

Figure 2
The structure for the compound valerophenone is shown in Figure 2.
(c)
Interpretation:
The structure for the compound
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
Answer to Problem 19.1P
The structure of
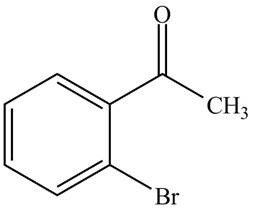
Explanation of Solution
The compound

Figure 3
The structure of the compound
(d)
Interpretation:
The structure for the compound
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
Answer to Problem 19.1P
The structure of the compound
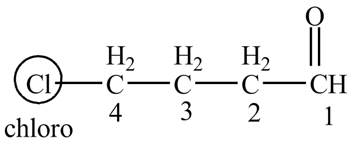
Explanation of Solution
The compound given in the question is

Figure 4
The IUPAC name of the compound is
The structure of the compound
(e)
Interpretation:
The structure for the compound
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
Answer to Problem 19.1P
The structure for the compound
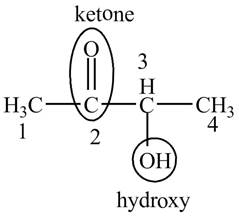
Explanation of Solution
The compound
This compound is also known as acetyl methyl carbinol. The structure of the compound is shown below.

Figure 5
The structure of the compound
(f)
Interpretation:
The structure for the compound
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name.
Answer to Problem 19.1P
The structure for the compound
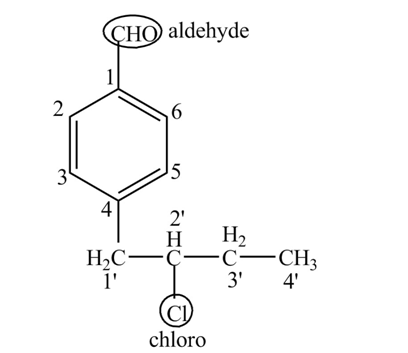
Explanation of Solution
The compound

Figure 6
The structure of the compound
(g)
Interpretation:
The structure for the compound
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name
Answer to Problem 19.1P
The structure for the compound
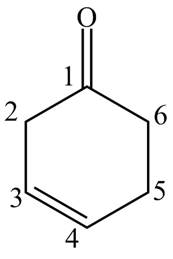
Explanation of Solution
The compound

Figure 7
The structure of the compound
(h)
Interpretation:
The structure for the compound
Concept introduction:
IUPAC stands for International Union of Pure and Applied Chemistry. The IUPAC gave certain rules which are used worldwide to name the organic compounds. Due to IUPAC naming, each compound has a unique name which is different from their common name
Answer to Problem 19.1P
The structure of the compound
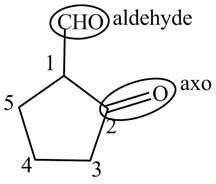
Explanation of Solution
The compound
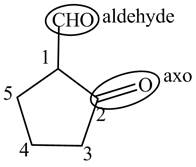
Figure 8
The structure of the compound
Want to see more full solutions like this?
Chapter 19 Solutions
Organic Chemistry
- A solution of acetone [(CH3)2C=O] in ethanol (CH3CH2OH) in the presence of a trace of acid was allowed to stand for several days, and a new compound of molecular formula C7H16O2 was formed. The IR spectrum showed only one major peak in the functional group region around 3000 cm−1, and the 1H NMR spectrum is given here. What is the structure of the product?arrow_forwardGive a systematic name for compound 2.1 including its stereochemical designationarrow_forwardDefine the Mechanism - Conversion of Carboxylic Acids to Amides with DCCarrow_forward
- A compound A, C7H12, was found to be optically active. On catalytic reduction over platinum catalyst, 2 equivalents of hydrogen were absorbed, yielding compound B, C7H16. On ozonolysis of A, two fragments were obtained. One fragment was identified as acetic acid. The other fragment, compound C, was an optically active carboxylic acid, C5H10O2. Write the reactions and draw structures for A, B and C.arrow_forwardIf 2,2‑dimethylbutane were subjected to such a reaction, how many different compounds (ignoring optical isomers) would be obtained?arrow_forwardTreatment of salicylaldehyde (2-hydroxybenzaldehyde) with bromine in glacial acetic acid at 0°C gives a compound with the molecular formula C7H4Br2O2, which is used as a topical fungicide and antibacterial agent. Propose a structural formula for this compoundarrow_forward
- Based on the following H NMR and IR spectrums, what is the structure of C7H6N2?arrow_forwardAspirin, or 2-acetoxybenzoic acid, (C9H8O4) is often synthesised from salicylic acid.(i) Sketch and discuss any changes in the number of possible structural conformations ofaspirin relative to those of salicylic acid. (ii) Re-draw the structure predicted to be the lowest energy conformation of aspirin,indicating any expected stabilising and destabilising interactions. Justify your choice.arrow_forward(a) Draw the structures of the following compounds :(i) 4-Chloropentan-2-one (ii) p-Nitropropiophenone(b) Give tests to distinguish between the following pairs of compounds :(i) Ethanal and Propanal (ii) Phenol and Benzoic acid(iii) Benzaldehyde and Acetophenonearrow_forward
- Compound A is an alcohol that undergoes oxidation to produce compound B.Compound B is a ketone that gives positive triiodomethane reaction. Compound B isthen reacted with phenyl magnesium bromide, C6H5MgBr in the presence of aqueousacid to form compound C. Compound C has the molecular formula of C9H12O. Deducethe structure for compound A, B and C. PLEASE PROVIDE CLEAR DRAWINGS AND EXPLANATIONSarrow_forwardwhen s-2-butanol is allowed to stand in dilute sulfuric acid for several days, it slowly forms a racemic mixture. why this happen and put illustratearrow_forwardDraw a line structure clearly showing the stereochemistry of (3S,4S)-4-hydroxy-3,5-dimethylhexanoic acid.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

