
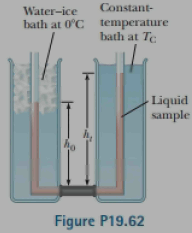
The measurement of the average coefficient of volume expansion β for a liquid is complicated because the container also changes size with temperature. Figure P19.62 shows a simple means for measuring β despite the expansion of the container. With this apparatus, one arm of a U-tube is maintained at 0°C in a water-ice bath, and the other arm is maintained at a different temperature Tc in a constant-temperature bath. The connecting tube is horizontal. A difference in the length or diameter of the tube between the two arms of the U-tube has no effect on the pressure balance at the bottom of the tube because the pressure depends only on the depth of the liquid. Derive an expression for β for the liquid in terms of h0, hi and Tc.
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
Physics for Scientists and Engineers, Technology Update (No access codes included)
- In Figure P19.22, the change in internal energy of a gas that is taken from A to C along the blue path is +800 J. The work done on the gas along the red path ABC is 500 J. (a) How much energy must be added to the system by heat as it goes from A through B to C? (b) If the pressure at point A is five times that of point C, what is the work done on the system in going from C to D? Figure P19.22 (c) What is the energy exchanged with the surroundings by heat as the gas goes from C to A along the green path? (d) If the change in internal energy in going from point D to point A is +500 J, how much energy must be added to the system by heat as it goes from point C to point D?arrow_forwardA rubber balloon is filled with 1 L of air at 1 atm and 300 K and is then put into a cryogenic refrigerator at 100 K. The rubber remains flexible as it cools. (i) What happens to the volume of the balloon? (a) It decreases to 13L. (b) It decreases to 1/3L. (c) It is constant. (d) It increases to 3L. (e) It increases to 3 L. (ii) What happens to the pressure of the air in the balloon? (a) It decreases to 13atm. (b) It decreases to 1/3atm. (c) It is constant. (d) It increases to 3atm. (e) It increases to 3 atm.arrow_forward(a) The inside of a hollow cylinder is maintained at a temperature Ta, and the outside is at a lower temperature, Tb (Fig. P19.45). The wall of the cylinder has a thermal conductivity k. Ignoring end effects, show that the rate of energy conduction from the inner surface to the outer surface in the radial direction is dQdt=2Lk[TaTbln(b/a)] Suggestions: The temperature gradient is dT/dr. A radial energy current passes through a concentric cylinder of area 2rL. (b) The passenger section of a jet airliner is in the shape of a cylindrical tube with a length of 35.0 m and an inner radius of 2.50 m. Its walls are lined with an insulating material 6.00 cm in thickness and having a thermal conductivity of 4.00 105 cal/s cm C. A heater must maintain the interior temperature at 25.0C while the outside temperature is 35.0C. What power must be supplied to the heater? Figure P19.45arrow_forward
- A fixed quantity of helium gas is placed in a sealed container, and it undergoes one of the two-step processes, either A→B→D or A→C→D, as shown. The volume is an integer multiple of the volume v0, and the pressure is an integer multiple of the pressure p0. The temperature of state A is 54.3∘C Part (a) What is the temperature, in degrees Celsius, of state B? Part (b) What is the temperature, in degrees Celsius, of state C? Part (c) What is the temperature, in degrees Celsius, of state D? Part (d) If the sealed container holds 0.342mol of helium gas and v0=2950cm3, then what is the value, in atmospheres, of p0?arrow_forwardA U.S. penny has a diameter of 1.9000 cmcm at 20.0 ∘C∘C. The coin is made of a metal alloy (mostly zinc) for which the coefficient of linear expansion is 2.6×10−5K−12.6×10−5K−1. Part A What would its diameter be on a hot day in Death Valley ( 47 ∘C∘C )? Express your answer using five significant figures. Part B On a cold night in the mountains of Greenland (-50 ∘C∘C )? Express your answer using five significant figures.arrow_forwardThe pressure in a constant-volume gas thermometer is 0.700 atm at 100°C and 0.512 atm at 0°C. (a) What is the temperature when the pressure is 0.0682 atm?____ °C(b) What is the pressure at 571°C?____ atm The New River Gorge bridge in West Virginia is a 518-m-long steel arch. How much will its length change between temperature extremes −19°C and 40°C? _____ cmarrow_forward
- 5.0 moles of Helium gas that occupies a volume of 0.400 m3 while at a temperature of 20.00 C undergoes an isobaric expansion to 0.600 m3. What is the final temperature of the gas? a. 1670 C b. 1120 C c. 1940 C d. 1010 C e. 1330 Carrow_forward(a) A tank contains one mole of nitrogen gas at a pressure of 6.15 atm and a temperature of 26.0°C. The tank (which has a fixed volume) is heated until the pressure inside triples. What is the final temperature of the gas? °C (b) A cylinder with a moveable piston contains one mole of nitrogen, again at a pressure of 6.15 atm and a temperature of 26.0°C. Now, the cylinder is heated so that both the pressure inside and the volume of the cylinder double. What is the final temperature of the gas? °Carrow_forwardn = 3.9 moles of an ideal gas are pumped into a chamber of volume V = 0.135 m3 50% Part (a) The initial pressure of the gas is 1 atm. What is the initial temperature (in K) of the gas? T = 421.76T = 421.8 ✔ Correct! 50% Part (b) The pressure of the gas is increased to 10 atm. Now what is the temperature (in K) of the gas?arrow_forward
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning




