
Concept explainers
(a)
Interpretation:
The skeletal structure of
Concept Introduction:
Fatty acids are long-chain
Unsaturated fatty acids can be defined as the long-chain fatty acids which have a long hydrocarbon chain with −COOH group. In unsaturated fatty acids, the carbon chain must have at least one double bond.
Answer to Problem 34P
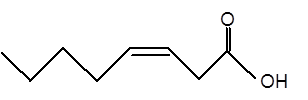
Explanation of Solution
Given:
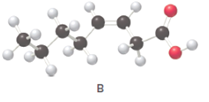
Fatty acids are long-chain carboxylic acids, which consist of two parts; long hydrocarbon chains and polar −COOH group. The long hydrocarbon chain consists of carbon and H atoms, so it has C-C and C-H bonds only, whereas, the polar −COOH group has polar C=O and C-O bonds. The unsaturated fatty acids have cis and trans-double bonds in the hydrocarbon chain.
In the ball-and-stick model, the red ball represents O atom, the black ball represents C atom and the white ball represents H atoms. Thus, the skeletal structure is:
(b)
Interpretation:
The omega-n designation for the
Concept Introduction:
Fatty acids are long-chain carboxylic acid, which may or may not have unsaturation in the molecule. They react with glycerol to form triglycerides. The reaction is called esterification as a carboxylic acid group reacts with the alcoholic group to form an ester group.
Unsaturated fatty acids can be defined as the long-chain fatty acids which have a long hydrocarbon chain with −COOH group. In unsaturated fatty acids, the carbon chain must have at least one double bond.
Answer to Problem 34P
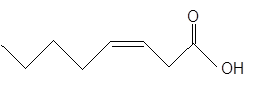
The
Explanation of Solution
Fatty acids are long-chain carboxylic acids, which consist of two parts; long hydrocarbon chains and polar −COOH group. If there is at least one double bond in the long hydrocarbon chain of the fatty acid, it is said to be an unsaturated fatty acid.
The unsaturated fatty acids can also classify as omega-n acids. Here, 'n' represents the position of the first
In the given fatty acid
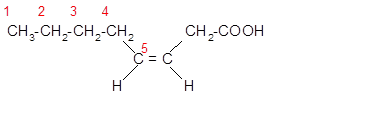
(c)
Interpretation:
The stereoisomer of the
Concept Introduction:
Fatty acids are long-chain carboxylic acid, which may or may not have unsaturation in the molecule. They react with glycerol to form triglycerides. The reaction is called esterification as a carboxylic acid group reacts with the alcoholic group to form an ester group.
Unsaturated fatty acids can be defined as the long-chain fatty acids which have a long hydrocarbon chain with −COOH group. In unsaturated fatty acids, the carbon chain must have at least one double bond.
Answer to Problem 34P
The stereoisomer of cis-fatty acid must be trans-isomer as given below;
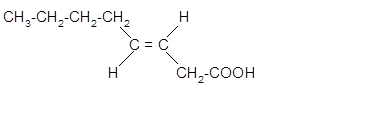
Explanation of Solution
Fatty acids are long-chain carboxylic acids, which consist of two parts; long hydrocarbon chains and polar −COOH group. The double bond can arrange in two ways, i.e., cis and Trans in the hydrocarbon chain of the fatty acid molecule. Hence, the stereoisomer of cis-fatty acid
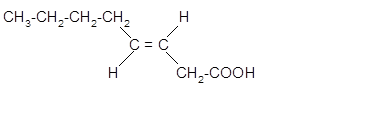
(d)
Interpretation:
The structure of wax formed by the reaction of
Concept Introduction:
Fatty acids are long-chain carboxylic acid, which may or may not have unsaturation in the molecule. They react with glycerol to form triglycerides. The reaction is called esterification as a carboxylic acid group reacts with the alcoholic group to form an ester group.
Unsaturated fatty acids can be defined as the long-chain fatty acids which have a long hydrocarbon chain with −COOH group. In unsaturated fatty acids, the carbon chain must have at least one double bond.
Answer to Problem 34P
The skeleton formula of wax formed by the reaction of
Explanation of Solution
Fatty acids are long-chain carboxylic acids, which consist of two parts; long hydrocarbon chains and polar −COOH group. Waxes are a good example of hydrolyzable lipids, which are composed of fatty acid and higher alcohols. They have an ester functional group which is formed between
Hence, the reaction of
Want to see more full solutions like this?
Chapter 19 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Which fatty acid is most likely to be a solid at room temperature? a. b b. c c. aarrow_forwardConsider we have a fatty acid that is described by 20:3 (8, 11, 14), what is its structure going to be? Draw its structure and identify what type of omega it is.arrow_forwardFatty acids are carboxylic acids that have long hydrocarbon chains attached to a carboxylate group. How does a saturated fatty acid differ from an unsaturated fatty acid? How are they similar?arrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





