
a)
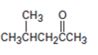
Interpretation:
How to prepare 4-methylpentan-2-one from 4-methyl-3-pentene-2-one is to be shown.
Concept introduction:
The double bond in an unsaturated
To show:
How to prepare 4-methylpentan-2-one from 4-methyl-3-pentene-2-one.
b)
Interpretation:
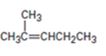
How to prepare 2-methylpent-2-ene from 4-methyl-3-pentene-2-one is to be shown.
Concept introduction:
The carbonyl group in a unsaturated ketone can be reduced to –CH2 without affecting the double bond by treating it with hydrazine in the presence of KOH (Wolf-Kishner reduction).
To show:
How to prepare 2-methylpent-2-ene from 4-methyl-3-pentene-2-one.
c)
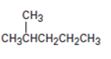
Interpretation:
How to prepare 2-methylpentane from 4-methyl-3-pentene-2-one is to be shown.
Concept introduction:
The double bond in an unsaturated ketone is reduced without affecting the carbonyl group by treating it with H2/Pd. The carbonyl group of the saturated ketone formed can be converted to –CH2 group by Wolff-Kishner reduction.
To show:
How to prepare 2-methylpentane from 4-methyl-3-pentene-2-one.
Trending nowThis is a popular solution!

Chapter 19 Solutions
Organic Chemistry
- Treatment of but-1-ene with hot KMno4/OH- gives product D, and subsequent acidification of the mixture gives product E. What are D and E. Give reaction equations.arrow_forwardCompound X, C7H15Cl, is a chiral product of the radical chlorination of 2-methylhexane.Base-promoted E2 elimination of X gives a single product.What is the structure of X?arrow_forwardPredict the major products of the following reactions.(a) 1@methylcyclohexene + aqueous Hg(OAc)2 (b) the product from part (a), treated with NaBH4arrow_forward
- What products would you expect from reaction of the following alkenes with NBS? If more than one product is formed, show the structures of all.arrow_forwardShow the products from the reaction of 2-methy-2-butene with d) HBr (no peroxide) e) Br2 f) O3 / Znarrow_forward2. Predict the major product from the following reaction sequence: (Write out complete equations showing the structure of all reactants and products.) (a) 3-methyl-1-hexene + 1)Hg(OAc)2, MeOH followed by 2) NaBH4 --------->arrow_forward
- Reaction of HBr with 2-methylpropene yields 2-bromopropane. What is the structure of the carbocation intermediate formed during the reaction? Show the mechanism of the reaction.arrow_forwardGive the following reaction A+3B --->2C.arrow_forward2) What is the main product for each of the following reactions? (d) Styrene + CH3CH2COCl AlCl3, then Br2, Fe -----> ? (e) Benzene + 1-chloro-2,2-dimethylpropane, AlCl3 -----> ? (h) Benzene + HCOCl, AlCl3 then 1-chlorobutane, AlCl3 -----> ?arrow_forward
- Show the product, including stereochemistry, of the reaction of the epoxide below with Na+ -CN, H2O.arrow_forward3H. Put it all together. Predict the E1 products of the following reaction. Label alkenes as E/Z. 4. Predict the E1 products of the following reaction. Label akenes as E/Z.arrow_forward(g) butan-2-one, CH3CH2COCH39-37 Show how you would synthesize the following compounds, starting with acetylene and any compounds containing nomore than four carbon atoms. 2,2-dibromohexanearrow_forward
