
Concept explainers
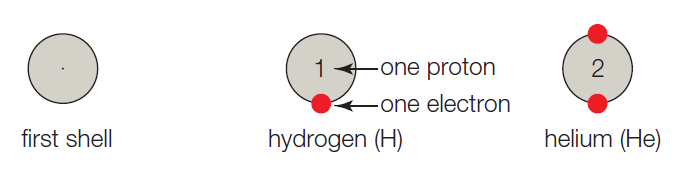
- A. The first shell corresponds to the first energy level, and it can hold up to 2 electrons. Hydrogen has one proton, so it has 1 electron and one vacancy. A helium atom has 2 protons, 2 electrons, and no vacancies.
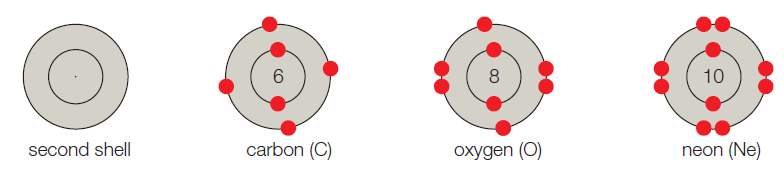
- B. The second shell corresponds to the second energy level, and it can hold up to 8 electrons. Carbon has 6 electrons, so its first shell is full. Its second shell has 4 electrons and four vacancies. Oxygen has 8 electrons and two vacancies. Neon has 10 electrons and no vacancies.
- C. The third shell corresponds to the third energy level, and it can hold up to 8 electrons. A sodium atom has 11 electrons, so its first two shells are full; the third shell has one electron. Thus, sodium has seven vacancies. Chlorine has 17 electrons and one vacancy. Argon has 18 electrons and no vacancies.
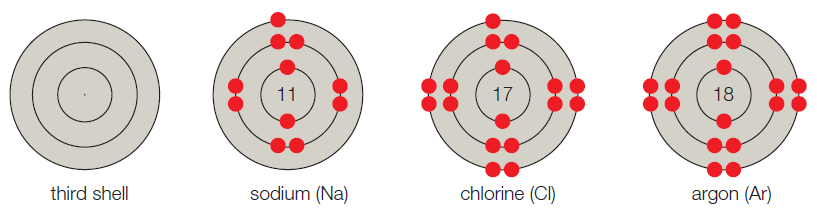
Figure It Out: Which of these models have unpaired electrons in their outer shell?
The atoms are referred to as building blocks of all substances. In other words, it is the basic fundamental unit of the matter. In every atom, the uncharged neutron and positively charged protons are present in the nucleus whereas the negatively charged electron moves around the nucleus. Generally, the shell model system is used to study the valence status of an atom. The atomic number is the number of protons present in the atom which determines the type of the atom
Explanation of Solution
The given shell model consists of about eight atoms namely helium, neon, hydrogen, oxygen, carbon, sodium, chloride, and argon. Of this given atom in the model the hydrogen, carbon, oxygen, chlorine and sodium have unpaired electrons in the outer shell. These unpaired electrons are available to pair with other unpaired electrons in another atom. Thus these atoms combine to form molecules.
Want to see more full solutions like this?
Chapter 2 Solutions
Biology Today and Tomorrow without Physiology (MindTap Course List)
- Magnesium has an atomic number of 12. Which of the following statements is true of a neutral magnesium atom? a. It has 12 protons, 12 electrons, and 12 neutrons. b. It has 12 protons, 12 electrons, and six neutrons. c. It has six protons, six electrons, and no neutrons. d. It has six protons, six electrons, and six neutrons.arrow_forwardOxygen (O) is a(n) _______; the oxygen we breathe (O2) is a(n) _______; and the carbon dioxide we exhale is a(n) _______. a. compound; molecule; element b. atom; compound; element c. element; atom; molecule d. atom; element; molecule e. element; molecule; compoundarrow_forwardWhich of the following statements about the mass number of an atom is incorrect? a. It has a unit defined as a dalton. b. On Earth, it equals the atomic weight. c. Unlike the atomic weight of an atom, it does not change when gravitational forces change. d. It equals the number of electrons in an atom. e. It is the sum of the protons and neutrons in the atomic nucleus.arrow_forward
- What atom has only one proton? a. hydrogen c. a free radical b. an isotope d. a radioisotopearrow_forwardA molecule of water is formed when one atom of oxygen is covalently bonded with two atoms of hydrogen. What is the total number of electron pairs that must be shared between the oxygen atom and the hydrogen atoms in order to complete the outer electron shell of all three atoms? a.2 b.3 c.4 d.1 e.5arrow_forward1.1. An atom of element X has an atomic number of 14 and a mass number of 30. Which of the following statements is correct? (A) An atom of sodium has 14 protons, 14 electrons, and 14 neutrons.(B) An atom of sodium has 14 protons, 16 electrons, and 14 neutrons(C) An atom of sodium has 14 protons, 14 electrons, and 16 neutrons.(D) An atom of sodium has 14 protons, 16 electrons, and 16 neutrons.1.2. The ability of carbon to form four covalent bonds with a variety of atoms is(A) Isomerism(B) Tetravalence(C) Hydrolysis(D) Quadravalence1.3. Water resists changing its temperature because of its … (A) High heat of vaporization(B) Low specific heat(C) High specific heat(D) Low heat of vaporization1.4. A solution where [H+] < [OH−] is more:(A) Acidic(B) Basic(C) Neutral(D) None of the above1.5. Malnourishment results when there is(A) A diet that does not provide enough fatty acids(B) A diet with short-term absence of one or more essential nutrients(C) A diet that does not provide enough…arrow_forward
- Oxygen has 8 protons, 8 neutrons, and 8 electrons. What is its atomic mass?(a) 8 (b) 16 (c) 24 (d) 32.arrow_forwardThe nucleus of an atom contains: a. neutrons and electrons b. protons and electrons c. protons only d. neutrons and protons e. neutrons onlyarrow_forward1. Which of the following is true of oxygen that has 8 protons, 8 neutrons, and 8 electrons? A. It has a charge of +8. B. It has a mass number of 8. C. It has an atomic number of 8. D. It has atomic number of 16. 2. Trace elements are those required by an organism in only minute quantities. Which of the following is a trace element that is required by all forms of life? A. arsenic B. iodine C. mercury D. ironarrow_forward
- A covalent bond forms whena. electrons are present in a valence shell.b. a valence electron is removed from one atom and added to another.c. a pair of valence electrons is shared between two atoms.d. the electronegativity of one atom is much greater than that ofanother atom.arrow_forwardCompared with 31P, the radioactive isotope 32P has(A) a different atomic number.(B) one more proton.(C) one more electron.(D) one more neutron.arrow_forward11H and 13H have (a) different chemical properties because they have different atomic numbers (b) the same chemical properties because they have the same number of valence electrons (c) different chemical properties because they differ in their number of protons and electrons (d) the samechemical properties because they have the same atomic mass (e) the same chemical properties because they have the same number of protons, electrons, and neutronsarrow_forward
 Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College
Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
 Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College




