
Concept explainers
Fill in the names beside the symbols of the following elements commonly found in living matter.
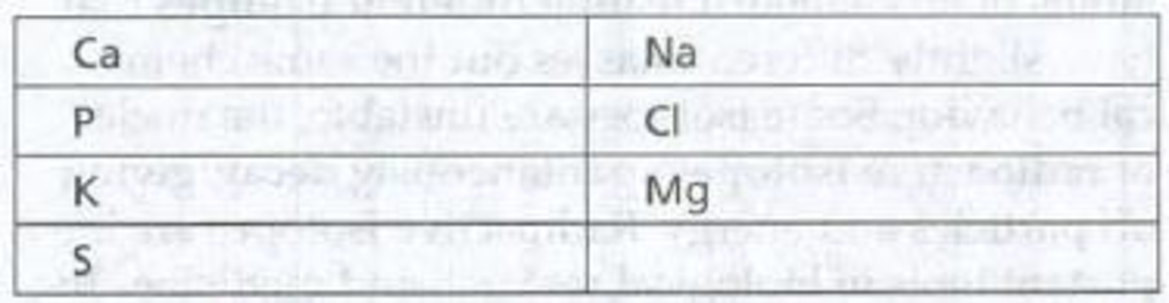
To fill: The names beside the symbol of the given element that are commonly found in living matter.
Introduction: In the simplest level of organization, the human body consists of various chemical structures. The substances that cannot be broken down into simpler substances by ordinary chemical reactions are called elements. Every element has its own chemical symbol.
Answer to Problem 1IQ
Tabular representation: The names of the symbol of the given element are in Table 1.
Table 1: Names of the symbol of the given element
| Ca -Calcium | Na- Sodium |
| P- Phosphorus | Cl- Chlorine |
| K- Potassium | Mg- Magnesium |
| S- Sulfur |
Explanation of Solution
Chemical elements are represented as symbols. In general, first one or two letters of element’s name are represented as a symbol of an element. For example, sulfur is represented as symbol “S” and calcium is represented as “Ca”. But few elements have symbols that have no relationship with their names. For example, potassium is represented by symbol “K” and sodium is represented as “Na”. For such cases, origin of the elements is used.
- Potassium- K- Latin name Kalium –means alkali
- Sodium- Na- Latin name for sodium carbonate- Natrium
Want to see more full solutions like this?
Chapter 2 Solutions
Study Guide for Campbell Biology
- Visit this website (http://openstaxcollege.org/l/ptable) to view the periodic table. In the periodic table of the elements, elements in a single column have the same number of electrons that can participate in a chemical reaction. These electrons are known as valence electrons. For example, the elements in the first column all have a single valence electron—an electron that can be donated in a chemical reaction with another atom. What is the meaning of a mass number shown in parentheses?arrow_forwardName two (2) elements which fit each of these descriptions Found in biological macromolecules Found in biological fluids in ionic form Trace elementsarrow_forwardDefine the following terms: a. metalloprotein b. primary structure c. secondary structure d. tertiary structure e. quaternary structurearrow_forward
- Define the following terms: a. glycoside b. disaccharide c. oligosaccharide d. polysaccharide e. glycosidic linkagearrow_forwardIf you took a sample of a particular element and found that one group had a specific number of neutrons, and another group had a different number of neutrons, those two groups are called ___ of each other. Isotopes Isomers anions cations What is the tube-like structure which allows direct access into the cytoplasm from the outside?arrow_forwardDefine the following terms:(a) Chiral (b) Achiral(c) Chiral carbon (d) Enantiomerarrow_forward
- Of the organic molecules listed below, which group has the greatest variety of three-dimensional shapes? fatty acids DNA phospholipids carbohydrates proteinsarrow_forwardThe triple point of an element is at 75 torr and 125°C. This means that liquid is more dense than solid cannot exist at 1 atmosphere pressure cannot have a vapor pressure less than 75 torr cannot exist above 125 degrees Celsius What is the electron geometry (or electron arrangement) around an atom in a molecule or ion which is surrounded by three lone pairs of electrons and two single bonds T-shaped see-saw trigonal planar trigonal bipyramidal linear A red blood cell placed in pure water will swell because water moves from a higer osmotic pressure to a lower osmotic pressure the osmotic pressure is greater in the cell than in the water the osmotic pressure is the same in the cell and the water the somotic pressure is greater in the water than in the cellarrow_forwardOxygen has 8 protons, 8 neutrons, and 8 electrons. What is its atomic mass?(a) 8 (b) 16 (c) 24 (d) 32.arrow_forward
- Define the following terms: a. carbonyl group b. carboxyl group c. amino group d. hydroxyl group e. peptide bondarrow_forwardA molecule of water is formed when one atom of oxygen is covalently bonded with two atoms of hydrogen. What is the total number of electron pairs that must be shared between the oxygen atom and the hydrogen atoms in order to complete the outer electron shell of all three atoms? a.2 b.3 c.4 d.1 e.5arrow_forwardDefine the following terms: a. dipole b. hydrogen bond c. electrostatic interaction d. covalent bond e. hydrationarrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College
Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College


