
Concept explainers
a. Rank the following alcohols from strongest to weakest acid:
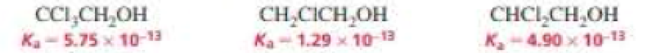
b. Explain the relative acidities.
(a)
Interpretation:
The given alcohols have to be ranked from strongest to weakest acid.
Concept introduction:
If a base receives one proton, then the formed species is a conjugate acid whereas an acid lose one proton, then the formed species is a conjugated base.
If an acid lose one proton, then the formed species is a conjugated base. Weak base forms stronger conjugated acid.
Acidity of species depends on the electronegativity of atom attached to the acidic proton. Order of electronegativity of hybridization is
Answer to Problem 47P
The given alcohols are ranked from strongest to weakest acid as follows,
Explanation of Solution
In hydrocarbons, if hydrogen atoms are replaced by electronegative atoms, it causes inductive electron withdrawal. It stabilizes its conjugate base thus increases the strength of the acid. The conjugated base of a weak acid is very strong. As the electronegativity of substituent increases, the greater will be the inductive electron withdrawal of the substituent making it a strong acid.
Therefore, the acidity order is:
The compound with three chlorine atoms near to the
(b)
Interpretation:
The relative acidities of the given alcohol compounds have to be explained briefly.
Concept introduction:
If a base receives one proton, then the formed species is a conjugate acid whereas an acid lose one proton, then the formed species is a conjugated base.
If an acid lose one proton, then the formed species is a conjugated base. Weak base forms stronger conjugated acid.
Acidity of species depends on the electronegativity of atom attached to the acidic proton. Order of electronegativity of hybridization is
Explanation of Solution
In hydrocarbons, if hydrogen atoms are replaced by electronegative atoms, it causes inductive electron withdrawal. It stabilizes its conjugate base thus increases the strength of the acid. The electron density near
Want to see more full solutions like this?
Chapter 2 Solutions
Organic Chemistry & Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
- Complete each Lewis structure, draw all important resonance structures, predict a value for thebond angles requested, and explain your reasoning. a. Nitrous acid (HNO2)HONOHON=ONO= b. Enolate ion (C2H3O) HC1C2=HC2C1=arrow_forwardRank the following compounds from strongest acid to weakest acid:arrow_forwardSort the compounds below in order of increasing acidity. Explain your answer.arrow_forward
- a) Circle, or where appropriate, draw in the hydrogen(s) on the compounds that are the most acidic. b) Briefly explain your answers for each.arrow_forwardRank the bases below in order of increasing basicity.arrow_forwardWhich is the most acidic among the ff. compounds: a) benzoic acid, b) benzamide c) methylbenzoate or d) benzoylchloride?arrow_forward
- The pKa of three CH bonds is given below.a.For each compound, draw the conjugate base, including all possible resonance structures. b. Explain the observed trend in pKaarrow_forwardWhat steps are needed to convert A to B?arrow_forwardCaftaric acid is found in grapes, wine, and raisins. Rank the labeledprotons in caftaric acid in order of increasing acidity.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
