
General Chemistry: Principles and Modern Applications (11th Edition)
11th Edition
ISBN: 9780132931281
Author: Ralph H. Petrucci, F. Geoffrey Herring, Jeffry D. Madura, Carey Bissonnette
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 20, Problem 11E
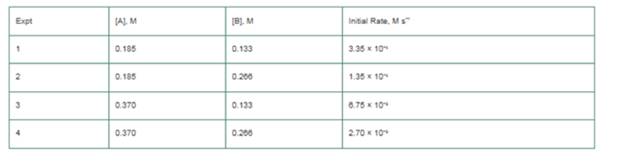
The initial
a. What is the order of reaction with respect to A and to B?
b. What is the overall reaction order?
c. What is the value of the rate constant, k?
Expert Solution & Answer
Learn your wayIncludes step-by-step video

schedule05:23
Chapter 20 Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
Ch. 20 - In the reaction 2A+BC+3D , reactant A is found to...Ch. 20 - From Figure 20-2 estimate the rate of reaction at...Ch. 20 - In the reaction A products, [A] is found to be...Ch. 20 - In the reaction A products, at t = 0. [A]= 0.1565...Ch. 20 - In the reaction A products. 4.40 min after the...Ch. 20 - Refer to Experiment 2 of Table 20.3 and to...Ch. 20 - For the reaction A+2BC ,the rate of reaction is...Ch. 20 - If the rate of reaction (20.3) is 5.7104 M s-1 ,...Ch. 20 - In the reaction A(g)B(g)+C(g) , the totalpressure...Ch. 20 - At 65C , the half-life for the first-order...
Ch. 20 - The initial rate of the reaction A+BC+D is...Ch. 20 - For the reaction A+BC+D , the following initial...Ch. 20 - Prob. 13ECh. 20 - The following data are obtained for the initial...Ch. 20 - One of the following statements is true and the...Ch. 20 - One of the following statements true and the other...Ch. 20 - The first-order reaction A products has t1/2=180...Ch. 20 - The reaction A products is first order in A....Ch. 20 - The reaction A products is first order A. a. If...Ch. 20 - In the first-order reaction A products, [A] =...Ch. 20 - In the first-order reaction A products, it found...Ch. 20 - The half-life of me radioactive isotope...Ch. 20 - Acetoacetic acid, CH2COOH2COOH , a reagent in...Ch. 20 - The following first-order reaction occurs in...Ch. 20 - For the reaction A- products, the following data...Ch. 20 - The decomposition of dimethyl ether at 504C is (...Ch. 20 - [Hint: There are several of arrivivg at answer for...Ch. 20 - [Hint: There are several of arrivivg at answer for...Ch. 20 - Prob. 29ECh. 20 - Prob. 30ECh. 20 - Prob. 31ECh. 20 - Prob. 32ECh. 20 - [Hint: There are several of arrivivg at answer for...Ch. 20 - [Hint: There are several ways of arrivivig at...Ch. 20 - Prob. 35ECh. 20 - Prob. 36ECh. 20 - For the reaction A products, the following data...Ch. 20 - Prob. 38ECh. 20 - For the reaction A products, the data tabulated...Ch. 20 - For the reaction A2B+C , the following data are...Ch. 20 - In three different experiments, the following...Ch. 20 - Ammonia decomposes on the surface of a hot...Ch. 20 - Prob. 43ECh. 20 - Consider three hypothetical reactions A — products...Ch. 20 - Prob. 45ECh. 20 - If even tiny sped is introduced into a mixture of...Ch. 20 - For me reversible reaction A+BC+D , the enthalpy...Ch. 20 - Prob. 48ECh. 20 - By inspection of the reaction profile for the...Ch. 20 - By inspection of the reaction profile for the...Ch. 20 - The rate constant for the reaction...Ch. 20 - At what temperature will the rate constant for the...Ch. 20 - Prob. 53ECh. 20 - The reaction C2H5+OHC2H5OH+I was studied in an...Ch. 20 - The first-order reaction A products has a...Ch. 20 - For the first-order reaction N2O4(g)2NO2+12O2g...Ch. 20 - Prob. 57ECh. 20 - Concerning the rule of thumb stated r Exercise 57,...Ch. 20 - The following statements about catalysis are not...Ch. 20 - Prob. 60ECh. 20 - What are the similarities and differences between...Ch. 20 - Certain gas-phase reactions on a heterogeneous...Ch. 20 - The graph show s the effect of enzyme...Ch. 20 - The graph shows the effect of temperature on...Ch. 20 - Prob. 65ECh. 20 - Prob. 66ECh. 20 - The reaction 2NO+2H2N2+2H2O is second order m [NO]...Ch. 20 - The mechanism proposed for me reaction of H2(g)...Ch. 20 - The reaction 2NO+Cl22NOCl has rate law: rate of...Ch. 20 - A simplified rate law 1o the reaction 2O2(g)3O2(g)...Ch. 20 - Prob. 71ECh. 20 - One proposed meachanism for the condensation of...Ch. 20 - Suppose that the reaction r Example 20-8 is first...Ch. 20 - [A]t as a function of time for the reaction A —...Ch. 20 - Exactly 300 s after decomposition of H2O2(aq)...Ch. 20 - Use the method of Exercise 75 to determine the...Ch. 20 - Prob. 77IAECh. 20 - Prob. 78IAECh. 20 - Hydroxide ion is involved in the mechanism of the...Ch. 20 - The half-life for the first-order decomposition of...Ch. 20 - The decomposition of ethylene oxide at 690 K is...Ch. 20 - Prob. 82IAECh. 20 - The following data are for the reaction 2 A + B ...Ch. 20 - Prob. 84IAECh. 20 - Prob. 85IAECh. 20 - Prob. 86IAECh. 20 - The following three-step mechanism has been...Ch. 20 - Prob. 88IAECh. 20 - Prob. 89IAECh. 20 - Prob. 90IAECh. 20 - Prob. 91IAECh. 20 - Prob. 92IAECh. 20 - Prob. 93IAECh. 20 - You want to test the following proposed mechanism...Ch. 20 - Prob. 95IAECh. 20 - Benzenediazonium chloride decomposes by a...Ch. 20 - The object is to study the kinetics of the...Ch. 20 - Prob. 98SAECh. 20 - Prob. 99SAECh. 20 - Explain the important distinctions between each...Ch. 20 - Prob. 101SAECh. 20 - A first-order reaction A — products, has a...Ch. 20 - Prob. 103SAECh. 20 - Prob. 104SAECh. 20 - The rate of a chemical reaction generally...Ch. 20 - For the reaction A+B2C, which proceeds by a...Ch. 20 - Prob. 107SAECh. 20 - Prob. 108SAECh. 20 - Prob. 109SAECh. 20 - For me reaction A products the following data are...Ch. 20 - For the reaction A+2BC+D , the rate law is rate...Ch. 20 - Prob. 112SAECh. 20 - If the plot of the reactant concentration versus...Ch. 20 - Prob. 114SAECh. 20 - Prob. 115SAE
Additional Science Textbook Solutions
Find more solutions based on key concepts
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach (4th Edition)
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
23. Give the symbol and name for (a) an isotope with a mass number of 37 and an atomic number of 17 and (b) an ...
Chemistry For Changing Times (14th Edition)
53. This reaction was monitored as a function of time:
A plot of In[A] versus time yields a straight ...
Chemistry: Structure and Properties
Several resonance structures are possible for each of the following ions. For each, drawthese resonance structu...
Inorganic Chemistry
Compare each of the mechanisms listed here with the mechanism for each of the two parts of the acid-catalyzed h...
Organic Chemistry (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Ammonia decomposes when heated according to the equation NH3(g) NH2(g) + H(g) The data in the table for this reaction were collected at a high temperature. Plot In [NH3] versus time and 1/[NH3] versus time. What is the order of this reaction with respect to NH3? Find the rate constant for the reaction from the slope.arrow_forwardNitrogen dioxide reacts with carbon monoxide by the overall equation NO2(g)+CO(g)NO(g)+CO2(g) At a particular temperature, the reaction is second order in NO2 and zero order in CO. The rate constant is 0.515 L/(mol s). How much heat energy evolves per second initially from 3.50 L of reaction mixture containing 0.0275 M NO2? See Appendix C for data. Assume the enthalpy change is constant with temperature.arrow_forwardA reaction of the form aA Products is second order with a rate constant of 0.169 L/(mol s). If the initial concentration of A is 0.159 mol/L, how many seconds would it take for the concentration of A to decrease to 5.50 103 mol/L?arrow_forward
- A study of the rate of dimerization of C4H6 gave the data shown in the table: 2C4H6C8H12 (a) Determine the average rate of dimerization between 0 s and 1600 s, and between 1600 s and 3200 s. (b) Estimate the instantaneous rate of dimerization at 3200 s from a graph of time versus [C4H6]. What are the units of this rate? (c) Determine the average rate of formation of C8H12 at 1600 s and the instantaneous rate of formation at 3200 s from the rates found in parts (a) and (b).arrow_forwardExperiments show that the reaction of nitrogen dioxide with fluorine, 2 NO2(g) + F2(g) —* 2 FNO2(g) has the rate law Rate = *[NO2][FJ The reaction is thought to occur in two steps. Step 1: NO2(g) + F,(g) —* FNO,(g) + F(g) Step 2: NO2(g) + F(g) — FNO2(g) Show that the sum of this sequence of reactions gives the balanced equation for the overall reaction. Which step is rate determining?arrow_forwardThe reaction I(aq)+OCl(aq)IO(aq)+Cl(aq) was studied, and the following data were obtained: [I]0(mol/L) [OCl]0(mol/L) Initial Rate (mol/L s) 0.12 0.18 7.91 102 0.060 0.18 3.95 102 0.030 0.090 9.88 103 0.24 0.090 7.91 102 a. What is the rate law? b. Calculate the value of the rate constant. c. Calculate the initial rate for an experiment where both Iand OCl are initially present at 0.15 mol/L.arrow_forward
- A The compound 1,3-butadiene (C4H6) forms 1,5-cyclooctadiene, C8H12 at higher temperatures. C4H6(g) C8H12(g) Use the following data to determine the order of the reaction and the rate constant, k. (Note that the total pressure is the pressure of the unreacted C4H6 at any time plus the pressure of the C8H12.)arrow_forwardThe reaction AB+C is known to be zero order in A and to have a rate constant of 5.0 102 mol/L s at 25C. An experiment was run at 25C where [A]0 = 1.0 103 M. a. Write the integrated rate law for this reaction. b. Calculate the half-life for the reaction. c. Calculate the concentration of B after 5.0 103 s has elapsed assuming [B]0 = 0.arrow_forwardIsomerization of CH3NC occurs slowly when CH3NC is heated. CH3NC(g) CH3CN(g) To study the rate of this reaction at 488 K, data on [CH3NC] were collected at various times. Analysis led to the following graph. (a) What is the rate law for this reaction? (b) What is the equation for the straight line in this graph? (c) Calculate the rate constant for this reaction. (d) How long does it take for half of the sample to isomerize? (e) What is the concentration of CH3NC after 1.0 104 s?arrow_forward
- From the following data, determine the rate equation, the rate constant, and the order with respect to A for the reaction A2C. [A](M) 1.33102 2.66102 3.99102 Rate (mol/L/h) 3.80107 1.52106 3.42106arrow_forwardKinetics II You and a friend are working together in order to obtain as much kinetic information as possible about the reaction A(g)B(g)+C(g). One thing you know before performing the experiments is that the reaction is zero order, first order, or second order with respect to A. Your friend goes off, runs the experiment, and brings back the following graph. a After studying the curve of the graph, she declares that the reaction is second order, with a corresponding rate law of Rate = k[A]2. Judging solely on the basis of the information presented in this plot, is she correct in her statement that the reaction must be second order? Here are some data collected from her experiment: Time (s) [A] 0.0 1.0 1.0 0.14 3.0 2.5 103 5.0 4.5 105 7.0 8.3 107 b The half-life of the reaction is 0.35 s. Do these data support the reaction being second order, or is it something else? Try to reach a conclusive answer without graphing the data. c What is the rate constant for the reaction? d The mechanism for this reaction is found to be a two-step process, with intermediates X and Y. The first step of the reaction is the rate-determining step. Write a possible mechanism for the reaction. e You perform additional experiments and find that the rate constant doubles in value when you increase the temperature by 10oC. Your lab partner doesnt understand why the rate constant changes in this manner. What could you say to your partner to help her understand? Feel free to use figures and pictures as part of your explanation.arrow_forwardThe decomposition of nitrogen dioxide at a high temperature NO2(g) NO(g) + O2(g) is second-order in this reactant. (a) Determine the rate constant for this reaction if it takes 1.76 min for the concentration of NO2 to fall from 0.250 mol/L to 0.100 mol/L (b) If the chemical equation is written as 2 NO2(g) 2 NO(g) + O2(g) what is the value of the rate constant?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
Kinetics: Chemistry's Demolition Derby - Crash Course Chemistry #32; Author: Crash Course;https://www.youtube.com/watch?v=7qOFtL3VEBc;License: Standard YouTube License, CC-BY