
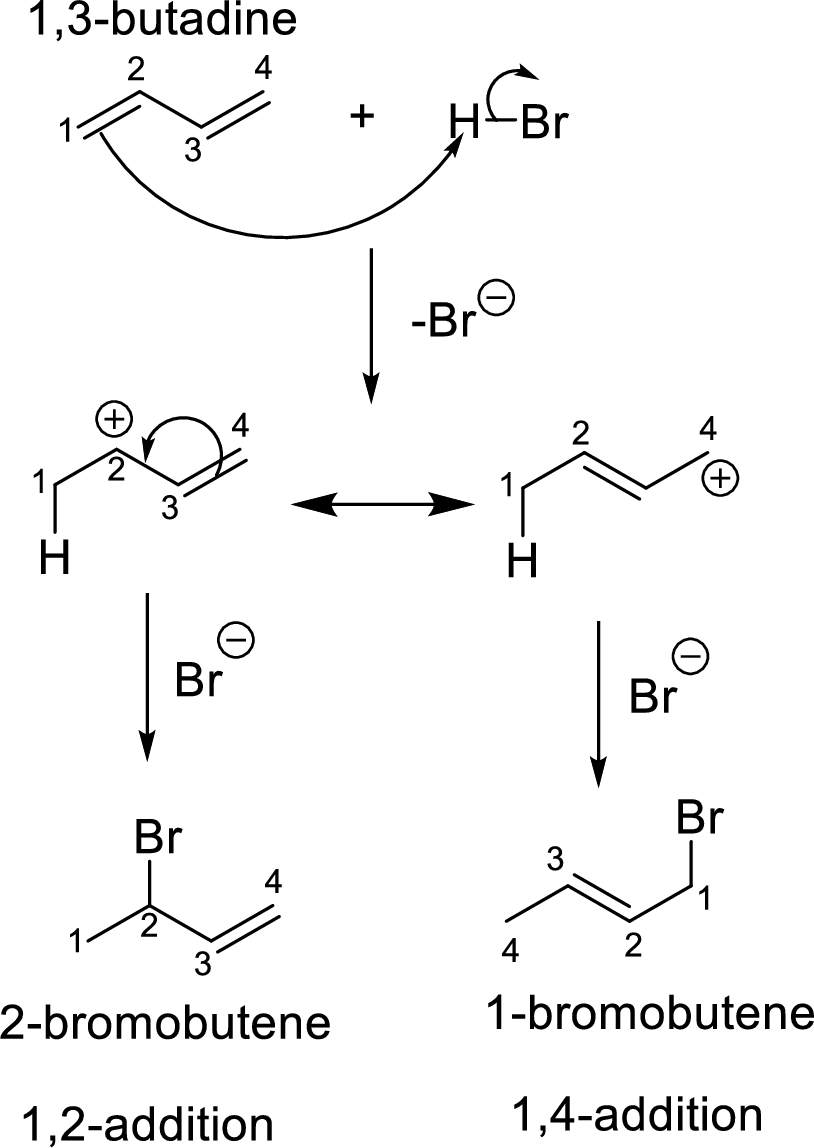
Interpretation: Among the given two molecules, the major product that is formed by
Concept Introduction:
Addition reaction of dienes:
The diene with a four carbon system is
Trending nowThis is a popular solution!

Chapter 20 Solutions
Organic Chemistry
- The 3D image below is that of an allylic carbocation intermediate formed by the protonation of a conjugated diene with HBr. Draw structural formulas for the final reaction products.arrow_forwardIgnoring isotope effects, after one E2 elimination with ethoxide, which bromine will predominate in the cyclohexene product? Explain your choice in 50 words or less.arrow_forwarddraw the expected transition state for the reaction of trans,trans -1,4 diphenyl butadiene with maleic anhydride using the endo rule and predict the stereochemistry of the major qnd minor products from the reaction. help me to draw this..arrow_forward
- Draw the structure of the conjugated diene that will react with one equivalent of HBr to yield a racemic mixture of 3-bromocyclohexene.arrow_forwardExclusive of stereoisomers, how many products are possible in the electrophilic addition of 1 mol of bromine to 2-methyl-1,3-butadiene?arrow_forwardProvide a reasonable synthetic strategy for the synthesis of a racemic mixture of (1R,2R) and (1S,2S)-2-bromo-1-methylcyclopentanol from methylcyclopentanearrow_forward
- The Wittig reaction can be used for the synthesis of conjugated dienes as the example below, 1-phenyl-penta-1, 3-diene. Propose two different sets of reagents that could be combined in a Wittig reaction to give 1-phenyl-1,3-pentadiene.arrow_forwardRank the following dienophiles in order of increasing reactivity towards 1,3- butadiene Please explainarrow_forwardwhat is the major monoalkylation product you would expect to obtain of bromobenzene from reaction of chloromethane and AlCl3?arrow_forward
- When carrying out this two step procedure (bromination and subsequent elimination) on cyclopentene, the lab students were surprised to find out that an alkyne was not the final product. What would the final product most likely be?arrow_forwardShow the process of synthesizing 2-methyl2-cyclohexenone from 2-methyl cyclohexanone. Write down the stages of the monocyclic reaction process in detail.arrow_forwardShow how free-radical halogenation might be used to synthesize the followingcompounds. In each case, explain why we expect to get a single major product.(a) 1-chloro-2,2-dimethylpropane (neopentyl chloride)arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

