
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 20, Problem 20.42P
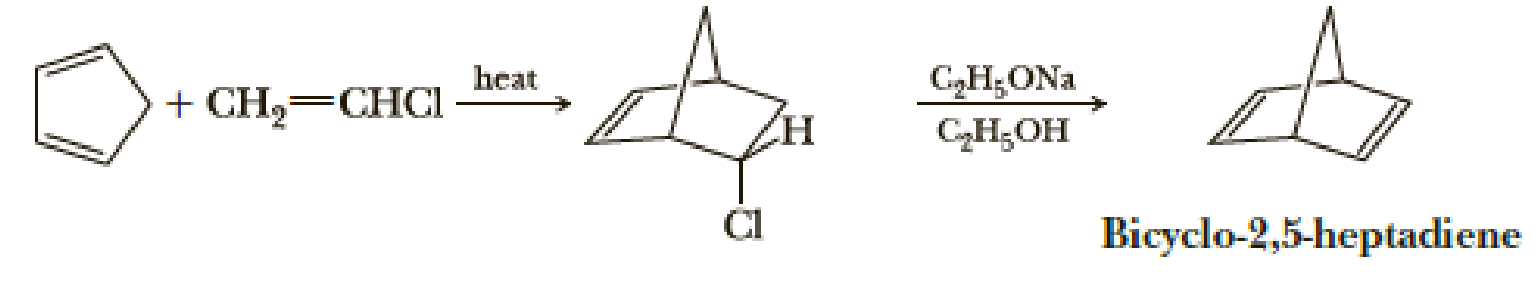
Bicyclo-2,5-heptadiene can be prepared in two steps from cyclopentadiene and vinyl chloride. Provide a mechanism for each step.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
When 1 mole of buta-1,3-diene reacts with 1 mole of HBr, both 3-bromobut-1-ene and 1-bromobut-2-ene are formed. Propose a mechanism to account for this mixture of products.
Treating 1-methyl-1-vinylcyclopentane with HCl gives mainly 1-chloro-1,2-dimethylcyclohexane.
Propose a mechanism for the formation of this product.
On treatment with HBr, vinylcyclohexane undergoes addition and rearrangement to yield 1-bromo-l-ethylcyclohexane. Using curved arrows, propose a mechanism to account for this result.
Chapter 20 Solutions
Organic Chemistry
Ch. 20.1 - Prob. 20.1PCh. 20.1 - Estimate the stabilization gained as a result of...Ch. 20.2 - Predict the product(s) formed by addition of one...Ch. 20.3 - Prob. 20.4PCh. 20.3 - Prob. 20.5PCh. 20.4 - Prob. 20.6PCh. 20.5 - Prob. 20.7PCh. 20.5 - Prob. 20.8PCh. 20.5 - Prob. 20.9PCh. 20.6 - Prob. 20.10P
Ch. 20.6 - Prob. 20.11PCh. 20.6 - Prob. 20.12PCh. 20 - If an electron is added to 1,3-butadiene, into...Ch. 20 - Prob. 20.15PCh. 20 - Predict the structure of the major product formed...Ch. 20 - Predict the major product formed by 1,4-addition...Ch. 20 - Predict the structure of the major 1,2-addition...Ch. 20 - Prob. 20.19PCh. 20 - Prob. 20.20PCh. 20 - Prob. 20.21PCh. 20 - Prob. 20.22PCh. 20 - Prob. 20.23PCh. 20 - Pyridine exhibits a UV transition of the type n at...Ch. 20 - Prob. 20.25PCh. 20 - Prob. 20.26PCh. 20 - Prob. 20.27PCh. 20 - Write the frontier molecular orbital analysis for...Ch. 20 - Prob. 20.29PCh. 20 - Draw structural formulas for the products of...Ch. 20 - Propose structural formulas for compounds A and B...Ch. 20 - Under certain conditions, 1,3-butadiene can...Ch. 20 - Prob. 20.33PCh. 20 - Prob. 20.34PCh. 20 - The following triene undergoes an intramolecular...Ch. 20 - Prob. 20.36PCh. 20 - Prob. 20.37PCh. 20 - Prob. 20.38PCh. 20 - Prob. 20.39PCh. 20 - The Diels-Alder reaction is not limited to making...Ch. 20 - The first step in a synthesis of dodecahedrane...Ch. 20 - Bicyclo-2,5-heptadiene can be prepared in two...Ch. 20 - Prob. 20.43PCh. 20 - Prob. 20.44PCh. 20 - Following is a retrosynthetic scheme for the...Ch. 20 - Prob. 20.46PCh. 20 - Prob. 20.47PCh. 20 - Prob. 20.48PCh. 20 - Prob. 20.49PCh. 20 - Prob. 20.50PCh. 20 - What reaction presented in this chapter is...Ch. 20 - Claisen rearrangement of an allyl phenyl ether...Ch. 20 - Prob. 20.53PCh. 20 - Prob. 20.54PCh. 20 - We now continue the use of organic chemistry...Ch. 20 - Write the products of the following sequences of...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Following is a retrosynthetic scheme for the synthesis of the tricyclic diene on the left. Show how to accomplish this synthesis from 2-bromopropane, cyclopentadiene, and 2-cyclohexenone.arrow_forwardNonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardBased on what you know about the relative stabilities of alkyl radicals and benzylic radicals, predict the product of addition of HBr to 1-phenylpropene in the presence of a free-radical initiator. Propose a mechanism for this reaction.arrow_forward
- Addition of HBr to 3,3-dimethyl-1-butene gives a mixture of two isomeric alkyl bromide products. Draw structures for the two products, and give a mechanistic explanation for their formation.arrow_forwardSometimes "Polyene" cyclization reactions can be catalyzed by a transition metal. Provide a mechanism for the reaction below.arrow_forwardIdentify two alkenes that react with HBr to form 1-bromo-1-methylcyclohexane without undergoing a carbocation rearrangement.arrow_forward
- What is the major product formed from reacting cyclohexene in aqueous Br2, followed by treatment with potassium tert-butoxide?arrow_forwardWhen sodium methoxide base and hydrogen bromide are removed from trans-1-bromo 2-methyl cyclohexane, considering the conformation of the starting compound, find the product that will be formed by writing and explaining the mechanism of the reaction.arrow_forwardAlkenes can be hydrated to form alcohols by (1) hydroboration followed by oxidation with alkaline hydrogen peroxide and (2) acid-catalyzed hydration. Compare the product formed from each alkene by sequence (1) with those formed from (2). Q.)Cyclopentenearrow_forward
- Following are example of Cope rearrangements of 1,5-dienes. Show that product can be formed in a single step by a mechanism involving redistribution of six electrons in a cyclic transition statearrow_forwardAs an example of a ver rare occurence, the following hydrocarbon reacts with two equivalents of butylithium to form a dianion of the formula [C8H6]2- . Propose a structure for this dianion and explain why this dianion forms so readily.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY