
Concept explainers
Interpretation:
The structural formula for each of the given compounds is to be drawn.
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done in such a way that the structure of organic compound is correctly interpreted from the name.
Rules for writing the structural formula from IUPAC are:
First identify the word root for the given compound.
The suffix used in the compound like –ane, ene, yne, ol, al and so on.
Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 28P
Solution:
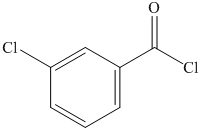
a) The structural formula of m-chlorobenzoyl chlorideis shown below.
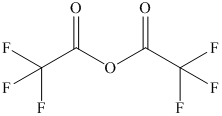
b) The structural formula of trifluoroacetic anhydride is shown below.
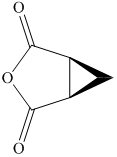
c) The structural formula of cis-
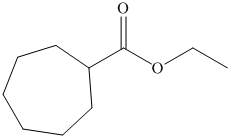
d) The structural formula of ethyl cycloheptanecarboxylateis shown below.
(e) The structural formula of
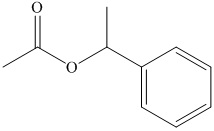
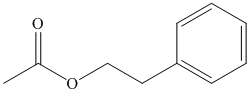
(f) The structural formula of
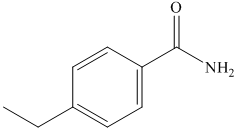
(g) The structural formula of p-ethylbenzamide is shown below.
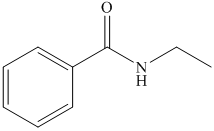
(h) The structural formula of N-ethylbenzamide is shown below.
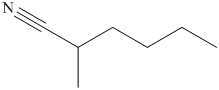
(i) The structural formula of
Explanation of Solution
a) m-Chlorobenzoyl chloride.
The name of compound suggests that chlorine group is present at meta position with respect to acyl group on benzene ring. The structure of compound is shown below.

b) Trifluoroacetic anhydride.
The name of the compound shows that anhydride group
group. The structure of compound is shown below.

c) cis-
The name of the compound indicates that two carboxylic groups are present on cyclopropane. Both carboxylic groups are present on the same plane. The structure of compound is shown below.

d) Ethyl cycloheptanecarboxylate
The name of the compound suggests that ester group is present in the structure. The cycloheptane ring is bonded to carbonyl carbon atom and ethyl group is bonded to oxygen atom. The structure of compound is shown below.

(e)
The name of the compound suggests that ester group is present in the structure. The methyl group is bonded to carbonyl carbon atom and ethyl group is bonded to oxygen atom. The structure of compound is shown below.

(f)
The name of the compound suggests that ester group is present in the structure. The methyl group is bonded to carbonyl carbon atom and ethyl group is bonded to oxygen atom. The second carbon atom is bonded to benzene ring. The structure of compound is shown below.

(g) p-Ethylbenzamide
The name of compound suggests that ethyl group is present at para position with respect to amide group on benzene ring. The structure of compound is shown below.

(h) N-Ethylbenzamide
The name of compound suggests that ethyl group is bonded to amide group which further bonded to benzene ring. The structure of compound is shown below.

(i)
The name of the compound indicates that it contains hydrocarbon chain of six carbon atoms. On the second carbon atom methyl group is present and on the first carbon atom nitrile group is present. The structure of compound is shown below.

Want to see more full solutions like this?
Chapter 20 Solutions
ORGANIC CHEMISTRY (LL)-W/ACCESS
- The following molecule belongs to a class of compounds called enediols; each carbon of the double bond carries an OH group. Draw structural formulas for the -hydroxyketone and the -hydroxyaldehyde with which this enediol is in equilibrium.arrow_forwardWhen benzaldehyde is added to the reagent prepared by reacting ethyl-2-bromoacetate with metallic zinc in benzene, which of the following molecules is obtained when the product is finally acidified.A. 3-phenyl-3-hydroxy-3-propanoate B. ethyl-3-phenylpropanoate C. ethyl-3-hydroxypropanoate D. ethyl-3-phenylpropanoate E ethyl-3-hydroxy-3-phenylpropanoatearrow_forwardWhich of the following lists functional groups in order of increasing reactivity towards nucleophiles? carboxylic acid < ester < acid anhydride < acid chloride ester < acid chloride < acid anhydride < carboxylic acid acid chloride < ester < carboxylic acid < acid anhydride carboxylic acid < ester < acid chloride < acid anhydride acid anhydride < acid chloride < ester < carboxylic acidarrow_forward
- Provide the structural formula of the products that are formed when acetophenone reacts with these reagents: 1. NaBH4 in Methanol 2. Ag(NH3)2+ 3. C6H5MgBr, then H3O+ 4. H2 in Pt 5. Hydroxylamine 6. Phenylhydrazine 7. LiAlH4 then water 8. HCN 9. Diethylamine 10. 2 CH3OH, HCl catalystarrow_forwardWhat will be the right combination of reagents to be required for the synthesis of the following compound? a. Ethanal + n-Propylamine b. Ethanal + isopropylamine c. Propanal + Ethylamine d. Ethylamine + Ethanal .arrow_forwardDraw the structure of the following: (a) p-chlorobenzoic anhydride (b) cis-3-Methylcyclohexanecarbonyl bromide (c) Ethyl 3-methylpentanoate (d) 3-chloro-N-ethyl-N-methylbenzamidearrow_forward
- What is the name of the product formed as a result of the reaction between benzylamine and ethylacrylate (ethylpropenoate)? A. N-benzylpropenamideB. N-phenylpropanamideC. N-phenylpropenamideD. Ethyl-3-aminobenzylpropanoateE. N-Ethyl-N-benzylpropanamidearrow_forwardPlease indicate which is the most acidic compound in each of the following pairs: thank you p-Nitrophenol and m-nitrophenol 4-Methylbenzoic acid and 4-chlorobenzoic acid Benzoic acid and 4-nitrobenzoic acidarrow_forwardIn the following pairs of compounds, which is the most acidic? Benzoic acid and 4-nitrobenzoic acid p-Nitrophenol and m-nitrophenol 4-Methylbenzoic acid and 4-chlorobenzoic acidarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


