
Interpretation:
The reagent has to be given for the conversion of A to Tamoxifen.
Concept introduction:
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (
Grignard reagent is reaction with carbonyl compound such as
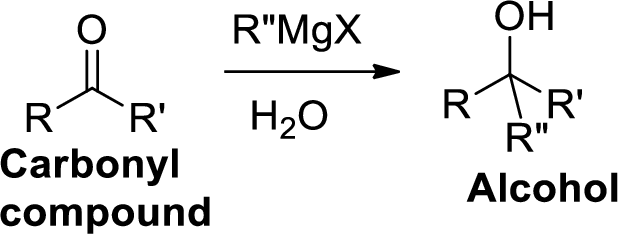
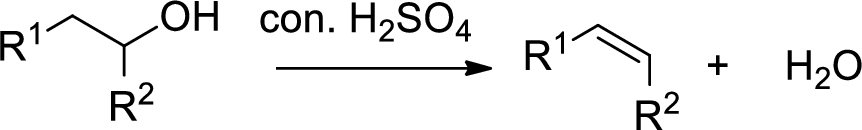
Dehydration reaction:
Removal of water molecule from the reaction when the alcohol is treated with strong acid like sulfuric acid.
Alcohol is reaction with concentrated sulfuric acid, first alcohol gets protonated forms carbocation (more stable carbocation) followed by elimination of proton (
Tertiary carbocation is more stable than the secondary, secondary carbocation is more stable than primary.
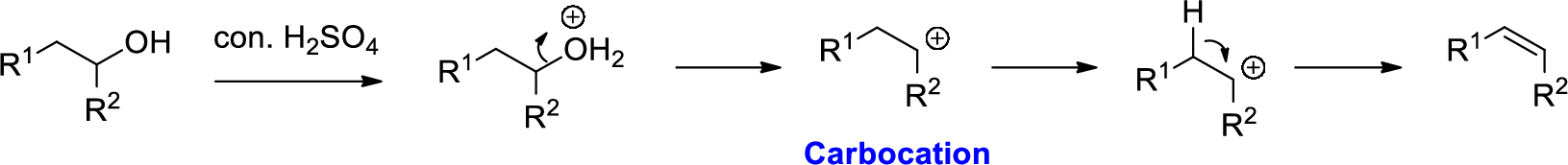
In dehydration reaction, sulfuric acid is act as a proton donor, and which is used to protonate the alcohol and makes carbocation therefore sulfuric acid is the driving force of the reaction. Dehydration reaction will not go without acid (sulfuric acid).
Want to see the full answer?
Check out a sample textbook solution
Chapter 21 Solutions
Organic Chemistry
- A newer generation of antipsychotics, among them clozapine, are now used to treat the symptoms of schizophrenia. These drugs are more effective than earlier drugs in improving patient response in the areas of social withdrawal, apathy, memory, comprehension, and judgment. They also produce fewer side effects such as seizures and tardive dyskinesia (involuntary body movements). In the following synthesis of clozapine, Step 1 is an Ullmann coupling, a type of nucleophilic aromatic substitution that uses a copper catalyst. (a) Show how you might bring about formation of the amide in Step 2. (b) Propose a reagent for Step 3. (c) Propose a mechanism for Step 4. (d) Is clozapine chiral? If so, how many of the possible stereoisomers are formed in this synthesis?arrow_forwardThe compound whose structure is shown here is acetyl acetone. It exists in two forms:the enol form and the keto form The molecule reacts with OH–to form an anion, [CH3COCHCOCH3] (often abbreviatedacac–for acetylacetonate ion). One or the most interesting aspects of this anion is thatone or more of them can react with transition metal cations to give stable, highlycolored compounds (a) Are the keto and enol forms of acetylacetone resonance forms? Explain youranswer.(b) What is the hybridization or each atom (except H) in the enol form? What changesin hybridization occur when it is transformed into the keto form?(c) What are the electron-pair geometry and molecular geometry around each C atomin the keto and enol forms? What changes in geometry occur when the keto formchanges to the enol form?(d) Draw three possible resonance structures for the acac–ion.(e) Is cis-trans isomerism possible in either the enol or the keto form of acetylacetone?(f) Is the enol form of acetylacetone polar?…arrow_forwardSubstituted phenols show substituent effects similiar to substituted benzoic acids. Should the pKa of phenol A, one of the naturally occurring phenols called urushiols isolated from poison ivy, be higher or lower than the pKa of phenol (C6H5OH, pKa = 10)? Explain.arrow_forward
- The compound acetophenone has a very similar molar mass to that of benzoic acid and benzamide. However, acetophenone has a much lower m.p. (20 °C) than both such that, by contrast, it is a liquid at room temperature. By considering intermolecular forces and comparing functional group structure, account for this big difference in physical properties.arrow_forwarda. Compound X is benzene, Y is acetic anhydride acid. Complete the following scheme and determine Z! b. Determine which reagents except acetic acid anhydrides can replace Y!arrow_forwardA diuretic is a compound that causes increased urination and thereby reduces fluid volume in the body. An important use of diuretics in clinical medicine is in the reduction of the fluid buildup, particularly in the lungs, that is associated with congestive heart failure. It is also used as an antihypertensive (i.e., to reduce blood pressure). Furosemide, an exceptionally potent diuretic, is prescribed under 30 or more trade names, the best known of which is Lasix. The synthesis of furosemide begins with treatment of 2,4-dichlorobenzoic acid with chlorosulfonic acid in a reaction called chlorosulfonation. The product of this reaction is then treated with ammonia followed by heating with furfurylamine. Q. Is furosemide chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forward
- Several additional amine syntheses are effectively limited to making primary amines. The reduction of azides and nitrocompounds and the Gabriel synthesis leave the carbon chain unchanged. Formation and reduction of a nitrile adds onecarbon atom. Show how these amine syntheses can be used for the following conversions. (c) 1@bromo@3@phenylheptane S 3@phenylheptan@1@amine (d) 1@bromo@3@phenylheptane S 4@phenyloctan@1@aminearrow_forwardDoes it safe to use this sunscreen K on acne prone skin. And can we rinse off it by only using facial gel mixed with water to rinse off it without use oil. Do I need to use micellar water for removing cosmetic before rinse off it with facial gel Sunscreen K has lists of ingredients below (descending order)…. AQUA DIMETHICONE CYCLOPENTASILOXANE PROPYLENE GLYCOL NIACINAMIDE DIETHYLHEXYL CARBONATE ZINC OXIDE CETEARYL ALCOHOL POLYMETHYL METHACRYLATE SODIUM POLYACRYLOYL-DIMETHYL TAURATE TITANIUM DIOXIDE ETHYLHEXYL METHOXYCINNAMATE BUTYL METHOXYDIBENZOYLMETHANE GLYCERYL STEARATE BIS-PEG/PPG-20/5 PEG/PPG-20/5 DIMETHICONE METHOXY PEG/PPG-25/4 DIMETHICONE HYDROGENATED POLYDECENE BUTYLENE GLYCOL OCTOCRYLENE PEG-40 STEARATE PHENOXYETHANOL HEXYL LAURATE POLYHYDROXYSTEARIC ACID CAPRYLIC/CAPRIC TRIGLYCERIDE STEARIC ACID CETYL ALCOHOL PEG-10 DIMETHICONE CETEARETH-20 TOCOPHERYL ACETATE STEARYL ALCOHOl PHENYL TRIMETHICONE TRIETHOXYCAPRYLYSILANE TRIDECETH-10 CHLORPHENESIN DISODIUM EDTA DIMETHICONOL…arrow_forwardCan you help with part b. Thank you!arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

