
Concept explainers
Problem 21.18
The
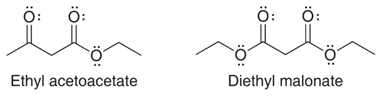
Interpretation:
The structural formulas for the most stable enolate of ethyl acetoacetate and diethyl malonate is to be drawn and by considering the resonance the delocalization of electrons in each enolate is to be shown.
Concept Introduction:
>Enol refers to an intermediate structure that consists of an
Resonance structure is a blend of two or more Lewis structures. Resonance structures are represented by a double headed arrow between the structures.
Trending nowThis is a popular solution!

Chapter 21 Solutions
Organic Chemistry - Standalone book
- Nabumetone is a pain reliever and anti-inflammatory agent sold under the brand name of Relafen. a. Write out a synthesis of nabumetone from ethyl acetoacetate. b. What ketone and alkyl halide are needed to synthesize nabumetone by direct enolate alkylation?arrow_forwardβ-Vetivone is isolated from vetiver, a perennial grass that yields a variety ofcompounds used in traditional eastern medicine, pest control, and fragrance. In one synthesis, ketone A is converted to β-vetivone by a two-step process: Michael reaction, followed by intramolecular aldol reaction. (a) What Michael acceptor is needed for the conjugate addition? (b) Draw a stepwise mechanism for the aldol reaction, which forms the six-membered ring.arrow_forwardDevise a synthesis of each compound from aniline (C6H5NH2) as starting material.arrow_forward
- Draw a stepwise mechanism for the conversion of lactone C to carboxylic acid D. C is a key intermediate in the synthesis of prostaglandins (Section 19.6) by Nobel Laureate E. J. Corey and co-workers at Harvard University.arrow_forwardβ-Vetivone is isolated from vetiver, a perennial grass that yields a variety of compounds used in traditional eastern medicine, pest control, and fragrance. In one synthesis, ketone A is converted to β-vetivone by a two-step process:Michael reaction, followed by intramolecular aldol reaction. (a) What Michael acceptor is needed for the conjugate addition? (See Problem 23.61 for another method to form the bicyclic ring system of β-vetivone.) (b) Draw a stepwise mechanism for the aldol reaction, which forms the six-membered ring.arrow_forwardDraw a stepwise mechanism for the following reactions, two steps in R. B. Woodward’s classic synthesis of reserpine in 1958. Reserpine, which is isolated from the extracts of the Indian snakeroot Rauwolfia serpentina Benth, was used at one time to manage mild hypertension associated with anxiety.arrow_forward
- Draw a stepwise mechanism for the following reactions, two steps in R. B. Woodward’s classic synthesis of reserpine in 1958. Reserpine, which is isolated from the extracts of the Indian snakeroot Rauwola serpentina Benth, was used at one time to manage mild hypertension associated with anxiety.arrow_forwardDevise a stepwise mechanism for the following reaction, a key step in the synthesis of the antibiotic abyssomicin C. Abyssomicin C was isolated from sediment collected from almost 1000 ft below the surface in the Sea of Japan.arrow_forwardDevise a synthesis of compound A from the given starting materials. Youmay use any other inorganic reagents or organic alcohols. A was used toprepare aliskiren, a drug used to treat hypertensionarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





