
Interpretation:
The given options should be identified that whether they are found in the structure of coenzyme FAD.
Concept Introduction:
Flavin Adenine Dinucleotide
Coenzyme: They are non-proteins molecules which helps enzymes for catalyzing the reaction.
Enzyme: The proteins that helps
ADP contains adenine attached to sugar backbone and 2 phosphate groups get attached to five carbon atoms present in ribose. The structure is as follows,
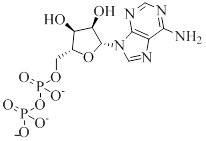
Phosphate anhydride bond: The bond that keeps phosphate groups and ribose together is termed as phosphate anhydride bond.
Benzene: The molecule that has 6 carbon atoms present in a ring to which each carbon has one hydrogen atom with it.
Want to see the full answer?
Check out a sample textbook solution
Chapter 21 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Given the following choices,a. what is the structure of the product of the reaction catalysed by ACAT?b. structure of the product produced from the reaction catalysed by phospolipase A2?c. general structure of TAGd. structure of glycerolarrow_forwardWhy is oxidative decarboxylation important?arrow_forwardName and draw the structure of the a-keto acid resulting when each of the following amino acids undergoes transamination with a-ketoglutarate: (a) aspartate, (b) glutamate, (c) alanine, (d) phenylalanine.arrow_forward
- With alanine AA, and using citric acid cycee and glycolysis, which Carbon atom would be labeled 1st with 14C n succinate? Why?arrow_forwardA reducing sugar will undergo the Fehling reaction, which requires a(potential) free aldehyde group. Which of the disaccharides shown inFigure are reducing and which are nonreducing?arrow_forwardThe fact that acid-anhydride bonds are less stable than ester bonds helps explain the fact that the bond between the: Beta- and gamma-phosphoryl groups in atp is a high energy bond, but not the bond between the alpha-phosphorly groups and the hydroxyl of ribose. Phosphorly groups and the hydroxyl at the C-3 position in 3-phosphoglycerate is a high-energy bond. Beta- gamma- phosphoryl groups in ATP is a high-energy bond, but not bond between the alpha and beta-phosphoryl groups. Alpha-phosphorly group (i.e, the one nearest the ribose) and the hydroxyl at the 5’ position in ATP is a high-energy bond.arrow_forward
- A reducing sugar will undergo the Fehling reaction, which requires a (potential) free aldehyde group. Which of the disaccharides as shown are reducing and which are nonreducing?arrow_forwardAssume you could make a trisaccharide by covalently joining xylose (C5H10O5) , xylose (C5H10O5) and fructose (C6H12O6). So 2x xylose, 1x fructose. What would be the chemical formula of this trisaccharide assuming regular dehydration synthesis as discussed in class? You do not need to worry about which carbons are being used for forming these glycosidic linkages.arrow_forwardAlcohol dehydrogenase, found in liver cells, converts ethanol into cetaldehyde. What type of protein is alcohol dehydrogenase?arrow_forward
- What are/Give the 4 structures of L-Cysteine from highly protonated to deprotonated form? What are the charges?arrow_forwardCarboxylic acids that undergo nucleophilic acyl substitutionreactions are often first converted to thioesters. For example,acetic acid forms thioester with a molecule called coenzyme A,which has a sulfhydryl group What is the leaving group in these reactions?arrow_forwardHow is ACP similar to coenzyme A? How is it different?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





