
Concept explainers
Interpretation:
The chirality centers in aldotetrose and ketopentose are to be calculated and the stereoisomers for each general case are to be determined.
Concept Introduction:
Carbohydrates are categorized mainly as monosaccharides, disaccharides, and polysaccharides. Monosaccharides are single sugar units, mainly glucose and fructose, disaccharides are two sugar units, such as sucrose, and polysaccharides are more than two sugar units, such as starch and cellulose.
Monosaccharides containing 3-carbon atoms are called triose, 4-carbon atoms called tetrose, 5-carbon atoms called pentose, and so on.
In chiral molecules, carbon atom having four nonidentical substituent groups is called the chirality center of that molecule. Chirality center may also be called stereocenter, which signifies any point in the molecule where the interchanging of any two groups may lead to stereoisomers. The carbon of a carbohydrate can be considered as chiral when the carbon has all four different substituents attached to it.
The stereoisomers are calculated as follows:
Here,
Answer to Problem 1PP
Solution:
a) Two
b) Two
c) Four
Explanation of Solution
a) The aldotetrose
A monosaccharide containing four carbon atoms is called a tetrose. An aldotetrose is a monosaccharide that contains
The structure of aldotetrose is as follows:
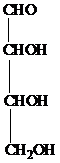
The carbon atom attached to four different groups is chiral carbon. The chiral center in ketopentose is marked by * as follows:
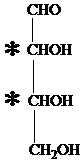
Hence, an aldotetrose has two chirality centers.
b) The ketopentose
A monosaccharide containing five carbon atoms is called a pentose. A pentose containing a keto group is called a ketopentose.
The structure of ketopentose is as follows:
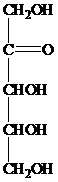
The carbon atom attached to four different groups is chiral carbon. The chiral center in ketopentose is marked by * as follows:
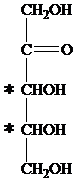
Hence, a ketopentose has two chirality centers.
c) The number of stereoisomers that will be expected from each general structure
Stereoisomers of a molecule have the same molecular formula, but different arrangement of atoms in space. Stereoisomers are different from enantiomers as they are not mirror images of each other, while enantiomers are mirror images of one another.
The compounds aldotetrose and ketopentose have two sets of enantiomers. The number of stereoisomers is calculated as:
Substitute 2 for
Hence, they will have four stereoisomers for each general structure.
Want to see more full solutions like this?
Chapter 22 Solutions
Organic Chemistry
- Choose the product that is expected when the β-pyranose form of compound A is treated with excess ethyl iodide in the presence of silver oxide. The following information can be used to determine the identity of compound A: 1. The molecular formula of compound A is C6H12O6.2. Compound A is a reducing sugar.3. When compound A is subjected to a Wohl degradation two times sequentially, D-erythrose is obtained.4. Compound A is epimeric with D-glucose at C3.5. The configuration at C2 is R.arrow_forwardDraw the product that is expected when the β-pyranose form of compound A is treated with excess ethyl iodide in the presence of silver oxide. The following information can be used to determine the identity of compound A: The molecular formula of compound A is C6H12O6. Compound A is reducing sugar. When compound A is subjected to a Wohl degradation two times sequentially, Derythrose is obtained. Compound A is epimeric with D-glucose at C3. The configuration at C2 is R.EXPLAIN IN DETAIL.arrow_forwardd-Xylose and d-lyxose are formed when d-threose undergoes a Kiliani–Fischer synthesis. d-Xylose is oxidized to an optically inactive aldaric acid,whereas d-lyxose forms an optically active aldaric acid. What are the structures of d-xylose and d-lyxose?arrow_forward
- Compound W shows stereoisomerism. Why are such isomers formed in approximately equimolar quantities no matter the synthetic pathway used for the preparation of the compound?arrow_forwardAn optically active D-aldopentose (A) produced an optically inactive alditol (B) upon treatment with H2/Pt. When the aldopentose (A) was subjected to a Ruff degradation, D-aldotetrose (C) was generated. The aldotetrose (C) gave an optically active aldaric acid (D) upon oxidation with HNO3. D-aldopentose (A) can be prepared from D-threose by a Killani Fischer synthesis. Propose structure of A through D.arrow_forwardA chiral amine A having the R conguration undergoes Hofmann elimination to form an alkene B as the major product. B is oxidatively cleaved with ozone, followed by CH3SCH3, to form CH2 = O and CH3CH2CH2CHO. What are the structures of A and B?arrow_forward
- Thalidomide is a chiral molecule and it was identified that the R-isomerproduced the sedative properties whereas the S-isomer produced the teratogenic effects.Identify the chiral centre in the thalidomide molecule and, using your knowledge of enolisation, illustrate mechanistically, and explain, why there would be no benefit to a patient taking the chirally pure R-isomer of the drug to avoid the sideeffects of the other enantiomer.arrow_forwardWhen the gum of the shrub Sterculia setigera is subjected to acidic hydrolysis, one of the water-soluble components of thehydrolysate is found to be tagatose. The following information is known about tagatose:(1) Molecular formula C6H12O6(2) Undergoes mutarotation.(3) Does not react with bromine water.(4) Reduces Tollens reagent to give d-galactonic acid and d-talonic acid.(5) Methylation of tagatose (using excess CH3 I and Ag2O) followed by acidic hydrolysis gives1,3,4,5-tetra-O-methyltagatose.(a) Draw a Fischer projection structure for the open-chain form of tagatose.(b) Draw the most stable conformation of the most stable cyclic hemiacetal form of tagatosearrow_forwardQuinapril (trade name Accupril) is used to treat high blood pressure andcongestive heart failure. One step in the synthesis of quinapril involvesreaction of the racemic alkyl bromide A with a single enantiomer of theamino ester B. Given the structure of quinapril, which one of these two products isneeded to synthesize the drug?arrow_forward
- The chiral hydroxyl compound P, C7H14O contains a five-membered ring and showspositive result in iodoform test. Compound P and sodium bromide can be produced byrefluxing compound Q with aqueous NaOH solution. Compound P is heated withbenzoic acid in the presence of concentrated H2SO4 to form a sweet-smelling product,compound R. Deduce the structure of P, Q and R. PLEASE PROVIDE CLEAR HAND WRITING AND SOLUTIONSarrow_forwardIs ganglefene chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forwarda. What is the classification of the Arabinose in terms of combined no. of carbons and highest functional group present? b. Provide the Cahn-Ingold-Prelog (R.S) Configuration of all the Chiral C present in the structure given above. c. State a Function of arabinose.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
