
Concept explainers
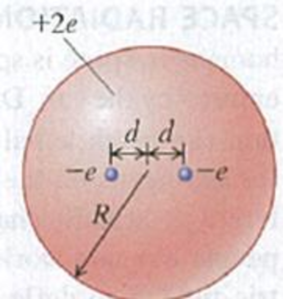
Thomson’s Model of the Atom, Continued. Using Thomson’s (outdated) model of (he atom described in Problem 22.50, consider an atom consisting of two electrons, each of charge −e, embedded in a sphere of charge +2e and radius R. In equilibrium, each electron is a distance d from the center of the atom (Fig. P22.51). Find the distanced in terms of the other properties of the atom.
Figure P22.51
Learn your wayIncludes step-by-step video

Chapter 22 Solutions
University Physics with Modern Physics, Volume 2 (Chs. 21-37); Mastering Physics with Pearson eText -- ValuePack Access Card (14th Edition)
Additional Science Textbook Solutions
Physics: Principles with Applications
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Lecture- Tutorials for Introductory Astronomy
- Compute for the ratio between the magnitudes of the repulsive force and gravitational force between two electrons. Consider the charge of an electron and its mass. a. 20 x10^(25) b. 4.2 x10^(42) c. 5.0 x10^(20) d. 6.25 x10^(18) ..... ..... .....arrow_forwardSay you have a point charge of one Coulomb and a hydrogen atom some distance away. Within what distance would the difference of forces on the proton and electron from the Coulomb charge exceed the attraction between the proton and electron that holds the hydrogen atom together. Treat the hydrogen atom as a proton and electron 0.53 × 10−10 m (a.k.a., one Bohr radius) apart.arrow_forwardWhat would be the total force (in nN) between two ions of substance1 when the distance between them is 0.236 nm? The attraction and repulsion energy coefficients between their ions are shown below (the respective energies are in ev units) A B Substance1 1.436 7.32E-06 8. Select one O a 21 Ob 1.42 OcCO O d -62 O e 9.5arrow_forward
- A cube structure having pv=(x^2)(y)(e^-z) [c/m^3] charge density distribution is being used to represent a discrete charge element Q. If edge of cube is 1 mm, answer the following question. m m im w n m w w w a) Derive Coulombs Law from Gauss Law. b) Calculate value of Q. c) If 2Q, 3Q, and -Q charges are placed on xy plane to (-1, –1,0), (2,0,0), (-3,1,0) points respectively, calculate the force due to electric field on a charge with 4Q at (1, -1,3) pointarrow_forwardTwo electrons in an atom are separated by 1.6 × 10^−10 m, the typical size of an atom. What is the force between them? The Coulomb constant is 9 × 10^9 N · m^2/C^2 . Answer in units of N.arrow_forward: In a box you find objects that come in three different colors: Green, Yellow, andPink. You’re told that each color corresponds to a charge state, either positive (+), negative (-), orneutral. You experiment and find that…Green is attracted to Yellow. Yellow is attracted to Pink Green is repelled by Green. Pink is repelled by Pink.What colors are the charged (either positive or negative) objects? What color is neutral object?arrow_forward
- Two protons (p) and two electrons (e) are arranged on a circle of radius = 5 [cm], with angles 0₁ = 10°, 0₂= 60°, 03= 15° and 04 = 45°, as shown in the figure. ( 9p = + 1.6 × 10-¹⁹ [C] and q = -1.6 × 10-¹⁹ [C]). The figure is not to scale. Р 03 04 a Find Enet, the net electric field vector produced at the center of the circle. Enet = ])i + ( ]) Ĵ [N/C] b. Where on the circle should a fifth point charge qo be placed (give its angle relative the +x-axis) and what is its value (calculate qo) in order to have Ēnet (the net electric field at the center of the circle) equals zero (Type the detailed solution to this question in the below box, Show all your calculation steps by typing in the box). Write an optional reasoning for your answer:arrow_forwardThe HCl molecule has a dipole moment of about 3.4x10-30 C·m. The two atoms areseparated by about 1.0x10-10 m.a. What is the net charge on each atom? Is this equal to an integer multiple of e? Ifnot, explain.b. What maximum torque would this dipole experience in a 2.5x104 N/C electricfield?c. How much energy would be needed to rotate one molecule 45° from itsequilibrium (lowest potential energy) position?arrow_forward6arrow_forward
- The net charge of an object can be found be summing the individual charges. Since charge is quantized, all net charges are just an integer multiple of the elementary charge. q =n e where q is the net charge and n is an integer. For example, sodium can be a +1 ion and chlorine can be a -1 ion. What does this mean concerning their charge and structure?arrow_forwardSuppose a capacitor consists of two coaxial thin cylindrical conductors. The inner cylinder of radius ra has a charge of +Q, while the outer cylinder of radius rp has charge -Q. The electric field E at a radial distance r from the central axis is given by the function: E = ae-T/ao + B/r + bo where alpha (a), beta (8), ao and bo are constants. Find an expression for its capacitance. First, let us derive the potential difference Vah between the two conductors. The potential difference is related to the electric field by: Vab = Edr = - Edr Calculating the antiderivative or indefinite integral, Vab = (-aage-r/a0 + B + bo By definition, the capacitance Cis related to the charge and potential difference by: C = Evaluating with the upper and lower limits of integration for Vab, then simplifying: C= Q/( (e-rb/ao - eTalao) + B In( ) + bo ( ))arrow_forwardA spherical shell of inner radius a cm has a volume charge density p(r, 0) =4/2tr cos 0 c/m³. The potential energy of this configuration is 5 cm and outer radius b = 7 Inner Yadius a=5 cm Outer radius b= 7 cm Volume charge dinsity pcr,@),4 cos0 .clm? Potential Energyarrow_forward
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

