
Concept explainers
(a)
Interpretation:
To explain why the given Lewis structure of CO is not acceptable.
Concept introduction:
Lewis structure is written by distributing the total valence electrons of each atoms involved in bonding, around each atom such that octet of each atom gets completed. Each electron is shown by a dot. If a positive charge is present on the ion then electrons equal to the charge on the ion must be subtracted from the total valence electrons and if a negative charge is present then that much electrons must be added to the total valence electrons.
Answer to Problem 22.56P
The given Lewis structure for
Explanation of Solution
In the given Lewis structure of CO the octet of O atom is complete but the octet of carbon atom is incomplete as shown below:
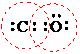
In the above structure around the oxygen atom there are eight electrons but around the carbon atom there are only six electrons that is the given structure does not follow the octet rule hence the given Lewis structure is not acceptable.
(b)
Interpretation:
The Lewis structure for the
Concept introduction:
Lewis structure is written by distributing the total valence electrons of each atoms involved in bonding, around each atom such that octet of each atom gets completed. Each electron is shown by a dot. If a positive charge is present on the ion, then electrons equal to the charge on the ion must be subtracted from the total valence electrons and if a negative charge is present then that much electrons must be added to the total valence electrons.
Answer to Problem 22.56P
The Lewis structure for

Explanation of Solution
The

In
The Lewis structure for

(c)
Interpretation:
To identify which electron pair is donated by the CO to the Fe metal for the formation of a coordinate covalent bond.
Concept introduction:
Lewis structure is written by distributing the total valence electrons of each atoms involved in bonding, around each atom such that octet of each atom gets completed. Each electron is shown by a dot. If a positive charge is present on the ion, then electrons equal to the charge on the ion must be subtracted from the total valence electrons and if a negative charge is present then that much electrons must be added to the total valence electrons.
Answer to Problem 22.56P
The lone pair of electron present on the C-atom donated to the Fe metal to form the coordinate covalent bond in
Explanation of Solution
In

(d)
Interpretation:
The Lewis structure for the
Concept introduction:
Lewis structure is written by distributing the total valence electrons of each atoms involved in bonding, around each atom such that octet of each atom gets completed. Each electron is shown by a dot. If a positive charge is present on the ion, then electrons equal to the charge on the ion must be subtracted from the total valence electrons and if a negative charge is present then that much electrons must be added to the total valence electrons.
Answer to Problem 22.56P
The Lewis structure for
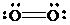
Explanation of Solution
The atomic number of O is 8 and its electronic configuration is

In
A coordinate covalent bond is formed when a donor molecule called as ligand donates a pair of electrons to the central metal atom. In case of
Want to see more full solutions like this?
Chapter 22 Solutions
Introduction to General, Organic and Biochemistry
- 22-62 Distinguish between intermolecular and intramolecular hydrogen bonding between backbone groups. Where in protein structures do you find one, and where do you find the other?arrow_forward22-53 Do iron and zinc ions play role in protein structure? If so, what is the role for either or both?arrow_forward22-73 Silver nitrate is sometimes put into the eyes of newborn infants as a preventive measure against gonorrhea. Silver is a heavy metal. Explain how this treatment may work against bacteria.arrow_forward
- 22-85 Denaturation is usually associated with transitions from helical structures to random coils. If an imaginary process were to transform the keratin in your hair from an (-helix to a (-pleated sheet structure, would you call the process denaturation? Explain.arrow_forward22-49 Based on your knowledge of the chemical properties of amino acid side chains, suggest a substitution for leucine in the primary structure of a protein that would probably not change the character of the protein very much.arrow_forward22-50 Is a random coil a (a) primary, (b) secondary, (c) tertiary or (d) quaternary structure? Explain.arrow_forward
- Give the functions of the following ingredients, then name a branded/commercial skin or hair care product where the said material is used. Ozokerite Wax Sodium Ascorbate Lactic Acid 90% Titanium Dioxide Magnesium Aluminum Silicate Mineral Oil 50 SUS Tween 80 Zinc Oxide Retinol DMDM HYDANTOINarrow_forwardWhat chemical is described as 1MHC2H3O2?arrow_forward22-72 What does the reducing agent do in straightening curly hair?arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning




