
Concept explainers
(a)
Interpretation:
Distribution of d-electrons and the geometry of the complex ion
Concept Introduction:
Complex compounds exist in following geometries - tetrahedral, square planar, octahedral etc.
When ligands approach the metal ion the degeneracy in d-orbitals of the metal ion is destroyed and they split into two different energy levels. In case of tetrahedral complex, the
In case of square planar complex, the 5 d-orbitals split into following pattern and it is given below with increasing order of energy.
(a)
Answer to Problem 22.60QP
The d-electrons are distributed in the complex ion
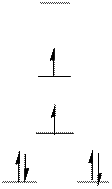
The complex has square planar geometry.
Explanation of Solution
In the complex ion

The distribution of d-electrons as shown above correlates to that of square planar geometry. Hence the complex ion
Splitting of d-orbitals determines the geometry of the complex.
(b)
Interpretation:
Distribution of d-electrons and the geometry of the complex ion
Concept Introduction:
Complex compounds exist in following geometries - tetrahedral, square planar, octahedral etc.
When ligands approach the metal ion the degeneracy in d-orbitals of the metal ion is destroyed and they split into two different energy levels. In case of tetrahedral complex, the
In case of square planar complex, the 5 d-orbitals split into following pattern and it is given below with increasing order of energy.
(b)
Answer to Problem 22.60QP
The d-electrons are distributed in the complex ion
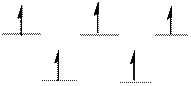
The complex has tetrahedral geometry.
Explanation of Solution
In the complex ion
Atomic number of Manganese is

The distribution of d-electrons as shown above correlates to that of tetrahedral geometry. Hence the complex ion
Splitting of d-orbitals determines the geometry of the complex.
(c)
Interpretation:
Distribution of d-electrons and the geometry of the complex ion
Concept Introduction:
Complex compounds exist in following geometries - tetrahedral, square planar, octahedral etc.
When ligands approach the metal ion the degeneracy in d-orbitals of the metal ion is destroyed and they split into two different energy levels. In case of tetrahedral complex, the
In case of square planar complex, the 5 d-orbitals split into following pattern and it is given below with increasing order of energy.
(c)
Answer to Problem 22.60QP
The d-electrons are distributed in the complex ion
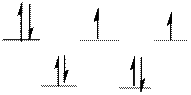
The complex has tetrahedral geometry.
Explanation of Solution
In the complex ion
Atomic number of Nickel is
The distribution of d-electrons as shown above correlates to that of tetrahedral geometry. Hence the complex ion
Splitting of d-orbitals determines the geometry of the complex.
(d)
Interpretation:
Distribution of d-electrons and the geometry of the complex ion
Concept Introduction:
Complex compounds exist in following geometries - tetrahedral, square planar, octahedral etc.
When ligands approach the metal ion the degeneracy in d-orbitals of the metal ion is destroyed and they split into two different energy levels. In case of tetrahedral complex, the
In case of square planar complex, the 5 d-orbitals split into following pattern and it is given below with increasing order of energy.
(d)
Answer to Problem 22.60QP
The d-electrons are distributed in the complex ion

The complex has square planar geometry.
Explanation of Solution
In the complex ion
Atomic number of Gold is

The distribution of d-electrons as shown above correlates to that of square planar geometry. Hence the complex ion
Splitting of d-orbitals determines the geometry of the complex.
Want to see more full solutions like this?
Chapter 22 Solutions
General Chemistry - Standalone book (MindTap Course List)
- Four different octahedral chromium coordination compounds exist that all have the same oxidation state for chromium and have H2O and Cl as the ligands and counterions. When 1 mole of each of the four compounds is dissolved in water, how many moles of silver chloride will precipitate upon addition of excess AgNO3?arrow_forwardExperiments show that K4[Cr(CN)6] is paramagnetic and has two unpaired electrons. The related complex K4[Cr(SCN)6] is paramagnetic and has four unpaired electrons. Account for the magnetism of each compound using the ligand field model. Predict where the SCN ion occurs in the spectro-chemical series relative to CN.arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





