
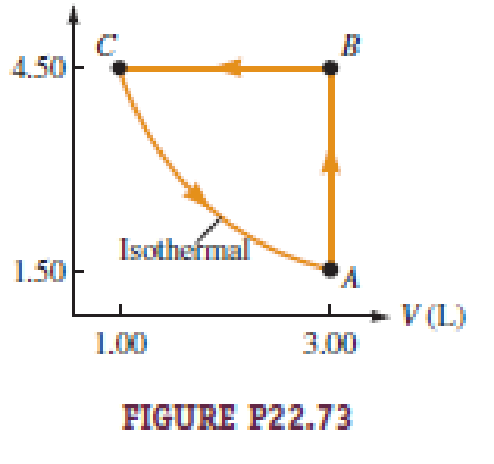
Figure P22.73 illustrates the cycle ABCA for a 2.00-mol sample of an ideal diatomic gas, where the process CA is a reversible isothermal expansion. What is a. the net work done by the gas during one cycle? b. How much energy is added to the gas by heat during one cycle? c. How much energy is exhausted from the gas by heat during one cycle? d. What is the efficiency of the cycle? e. What would be the efficiency of a Carnot engine operated between the temperatures at points A and B during each cycle?
(a)
The net work done by the gas during one cycle.
Answer to Problem 73PQ
The net work done by the gas during one cycle is
Explanation of Solution
The work done by the gas along AB is
Write the expression to calculate the work done by the gas along BC.
Here,
Write the expression to calculate the work done by the gas along CA.
Here,
Write the expression to calculate the total work done by the gas.
Here, W is the total work done by the gas.
Conclusion:
Substitute
Substitute
Substitute
Therefore, the net work done by the gas during one cycle is
(b)
The heat added in one cycle.
Answer to Problem 73PQ
The heat added in one cycle is
Explanation of Solution
For the diatomic gases, the specific heat capacity at constant volume is
The temperature at A and C of the gas is same.
Write the expression to calculate the temperature of the gas at A and C.
Here, T is the temperature of the gas at A and C, P is the pressure at A, V is the pressure at A n is the number of moles and R is the universal gas constant.
Write the expression to calculate the temperature at B.
Here,
Write the expression to calculate the energy added to the system.
Here, Q is the energy added to the system,
Substitute
Conclusion:
Substitute
Substitute
Substitute
Therefore, the heat added in one cycle is
(c)
The energy exhausted from the gas.
Answer to Problem 73PQ
The energy exhausted from the gas is
Explanation of Solution
For the diatomic gases, the specific heat capacity at constant pressure is
Write the expression to calculate the heat exhausted from the gas.
Here,
Substitute
Conclusion:
Substitute
Therefore, the energy exhausted from the gas is
(d)
The efficiency of the cycle.
Answer to Problem 73PQ
The efficiency of the cycle is
Explanation of Solution
Write the expression to calculate the efficiency of one cycle.
Here, e is the efficiency.
Conclusion:
Substitute
Therefore, the efficiency of the cycle is
(e)
The efficiency of the Carnot engine.
Answer to Problem 73PQ
The efficiency of the Carnot engine is
Explanation of Solution
Write the expression to calculate the efficiency of the Carnot engine.
Here,
Conclusion:
Substitute
Therefore, the efficiency of the Carnot engine is
Want to see more full solutions like this?
Chapter 22 Solutions
Physics for Scientists and Engineers: Foundations and Connections
- A 1.00-mol sample of a monatomic ideal gas is taken through the cycle shown in Figure P22.76. At point A, the pressure, volume, and temperature are Pi, Vi, and Ti, respectively. In terms of R and Ti, find (a) the total energy entering the system by heat per cycle, (b) the total energy leaving the system by heat per cycle, and (c) the efficiency of an engine operating in this cycle. (d) Explain how the efficiency compares with that of an engine operating in a Carnot cycle between the same temperature extremes.arrow_forwardA thermodynamic cycle is shown in Figure P21.34 for a gas in a piston. The system changes states along the path ABCA. a. What is the total work done by the gas during this cycle? b. How much heat is transferred? Does heat flow into or out of the system? Figure P21.34arrow_forwardFigure P21.36 shows a cyclic thermodynamic process ABCA for an ideal gas. a. What is the net energy transferred into the system by heat during each cycle? b. What would be the net energy transferred into the system by heat if the cycle followed the path ACBA instead? FIGURE P21.36 FIGURE P21.37arrow_forward
- (a) In reaching equilibrium, how much heat transfer occurs from 1.00 kg of water at 40.0C when it is placed in contact with 1.00 kg of 20.0C water in reaching equilibrium? (b) What is the change in entropy due to this heat transfer? (c) How much work is made unavailable, taking the lowest temperature to be 20.0C ? Explicitly show how you follow the steps in the Problem-Solving Strategies for Entropy.arrow_forwardA 1.00-mol sample of an ideal monatomic gas is taken through the cycle shown in Figure P21.37. The process A B is a reversible isothermal expansion. Calculate (a) the net work done by the gas, (b) the energy added to the gas by heat, (c) the energy exhausted from the gas by heat, and (d) the efficiency of the cycle. (e) Explain how the efficiency compares with that of a Carnot engine operating between the same temperature extremes. Figure P21.37arrow_forward(a) What is the hot reservoir temperature of a Carnot engine that has an eficiency of 42.0% and a cold reservoir temperature of 210C ? (b) What must the hot reservoir temperature be for a real heat engine that achieves 0.700 of the maximum eficiency, but still has an efficiency of 42.0% (and a cold reservoir at 27.0C )? (c) Does your answer imply practical limits to the efficiency of car gasoline engines?arrow_forward
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning





