
Concept explainers
(a)
Interpretation:
The structure of the product has to be drawn when the
Concept Introduction:
A primary alcohol gives
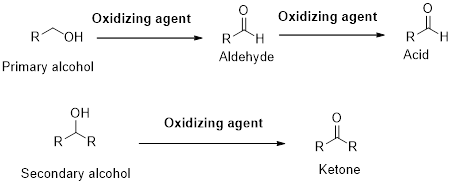
(b)
Interpretation:
The structure of the product has to be drawn when the
Concept Introduction:
A primary alcohol gives carboxylic acid

(c)
Interpretation:
The structure of the product has to be drawn when the
Concept Introduction:
A primary alcohol gives carboxylic acid

(d)
Interpretation:
The structure of the product has to be drawn when the
Concept Introduction:
A primary alcohol gives carboxylic acid

Trending nowThis is a popular solution!

Chapter 23 Solutions
Chemistry & Chemical Reactivity
- Complete the following chemical reactions. Make sure to draw and name all structural formulae for all organic compounds (both reactants and products). a. Oxidation of 2-pentanol. (K2Cr2O7/H+) → b. Formic acid + Isopropyl alcohol (2-propanol) → c. Acetic acid + Propylamine → d. Dehydration of 2,3-dimethyl-1-butanol (H+/heat) →arrow_forwardAcetyl chloride, CH3COCl, reacts with the hydroxyl groupsof alcohols to form ester groups with the elimination ofHCl. When an unknown compound X with formulaC4H8O3 reacted with acetyl chloride, a new compound Ywith formula C8H12O5 was formed.(a) How many hydroxyl groups were there in X?(b) Assume that X is an aldehyde. Write a possible structure for X and a possible structure for Y consistent with your structure for X.arrow_forwardWrite two complete, balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures:(a) ethanol reacts with propionic acid(b) benzoic acid, C6H5CO2H, is added to a solution of sodium hydroxidearrow_forward
- 1) The acid-catalyzed dehydration of 2-methyl-2-butanol yields two alkene products, what are the names of the two alkenes? 2) which of the two alkenes is the major product?arrow_forwarda) Draw the structure for ethyl 3-methyl butanoate. b) Which functional group(s) does this molecule contain? c) Which type of reaction could be used to synthesize this molecule?arrow_forwardDraw all alcohols with the formula C5H12O.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





