
Concept explainers
E. J. Corey’s 1964 total synthesis of α-caryophyllene (essence of cloves) solves a number of problems of construction of unusual-sized rings.
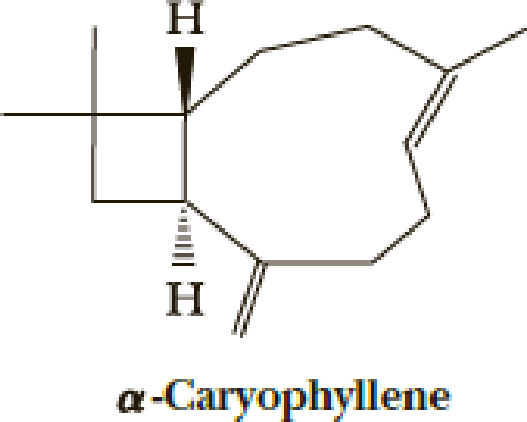
The first step uses an efficient photochemical [2 + 2] reaction. The desired stereochemistry and regiochemistry had been predicted based on model reactions.
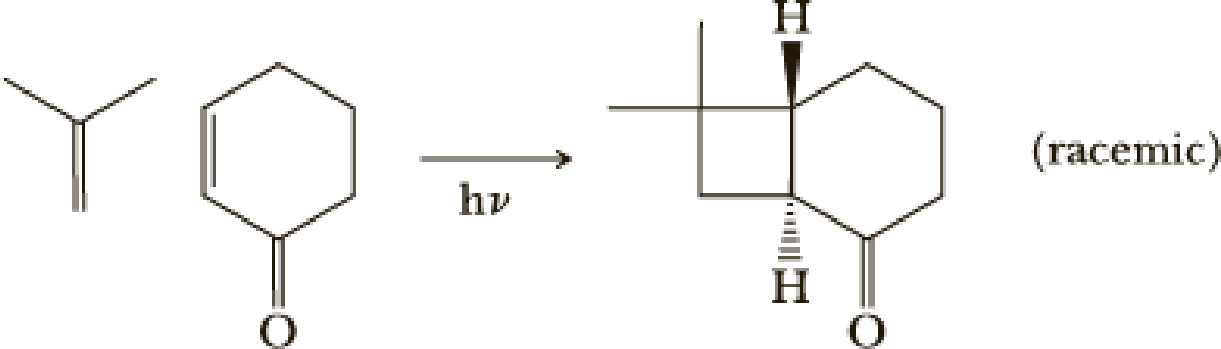
- (a) [2 + 2] Reactions are quite common in photochemical reactions. Would this reaction be predicted to occur in the ground state?
The next steps follow. Basic alumina is a chromatography support that will often act as a base catalyst.
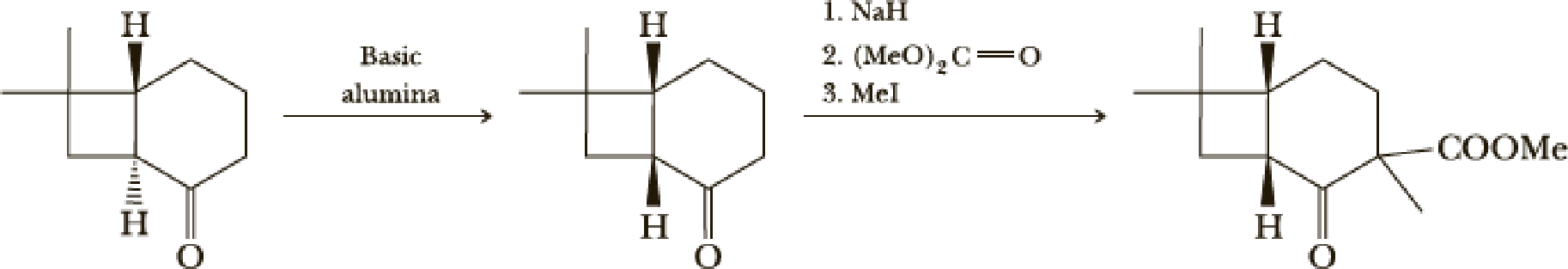
- (b) What is the mechanism of the first step?
- (c) What is the mechanism of the second step?
- (d) Look at later steps in the synthesis. Does the stereochemistry of the added carbomethoxy group matter?
The next steps are shown here.
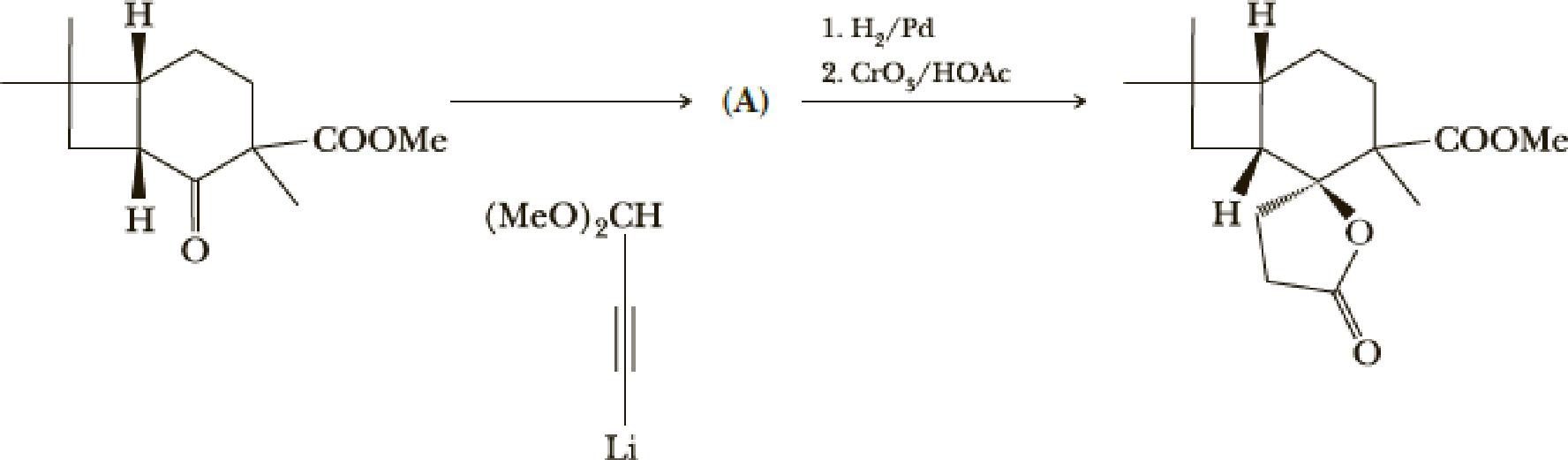
- (e) What is the structure of compound (A)?
- (f) Give a mechanism for the formation of the cyclized product.
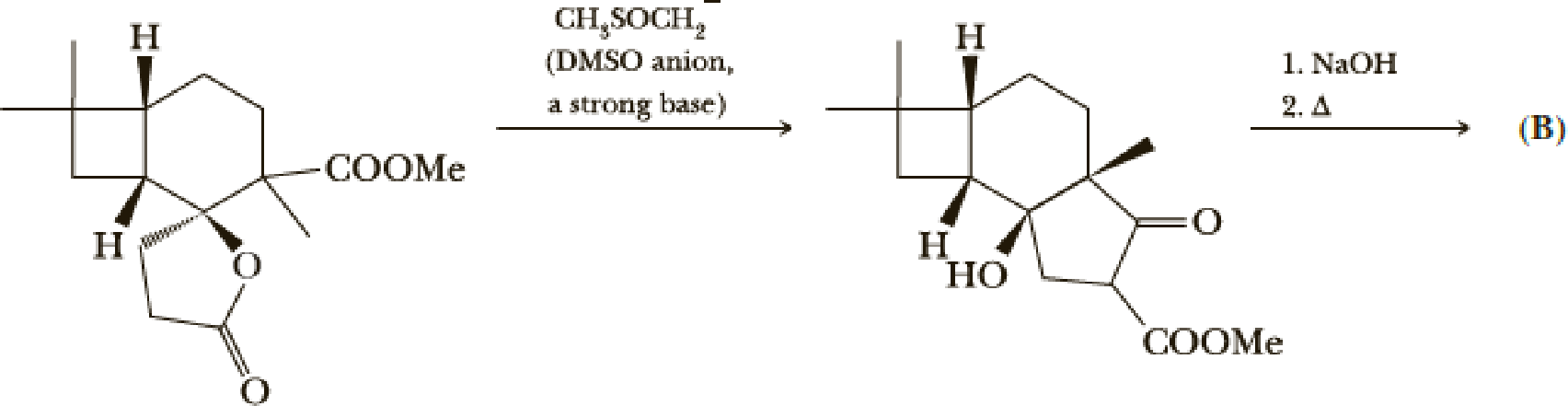
- (g) Give a mechanism for the first step. Hint: Attack on the lactone carbonyl may be the first step.
- (h) Give a structure for product (B).
The following two steps are next.
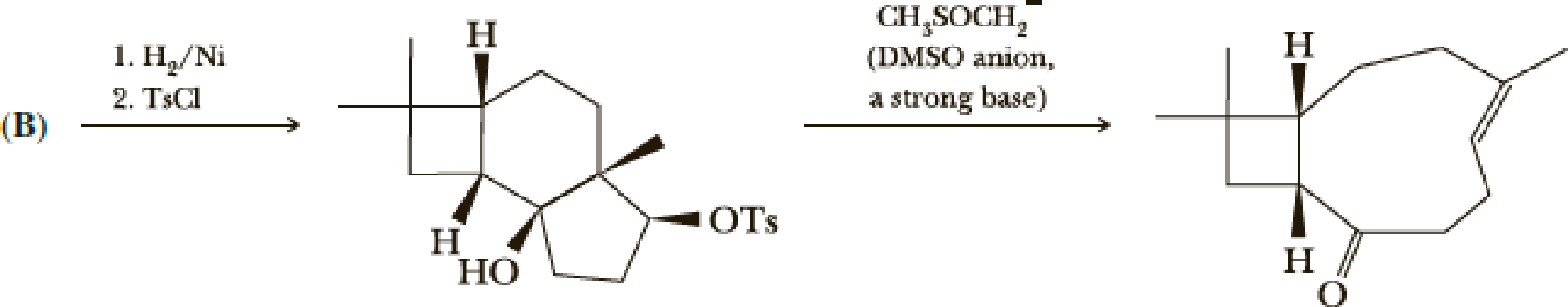
- (i) Show the reactions of (B).
- (j) Write a mechanism for the ring-opening reaction. Hint: Note the presence of an acidic proton and a good leaving group in the molecule.
The synthesis was completed by the following steps.
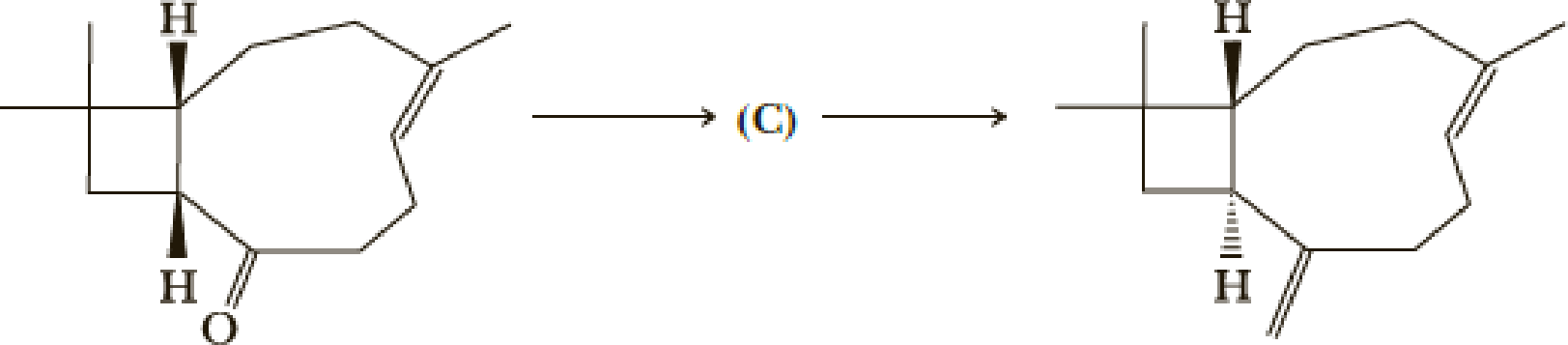
- (k) What is (C)?
- (l) What reagents would you use for these transformations?
(a)
Interpretation:
Whether the
Concept introduction:
Woodward-Hoffmann’s rule:
These are set of rules to determine the geometry and the feasibility of pericyclic reactions. The rules are as follow,
In an open chain system containing
In an open chain system containing
For photochemical condition the electron of HOMO is excited to LUMO and hence the reversal of the terminal symmetry relationship occurs.
Explanation of Solution
According to Woodward Hoffmann’s rule ground state
Ground state it shows
Hence ground state reaction is forbidden.
(b)
Interpretation:
The mechanism of the given step has to be given.
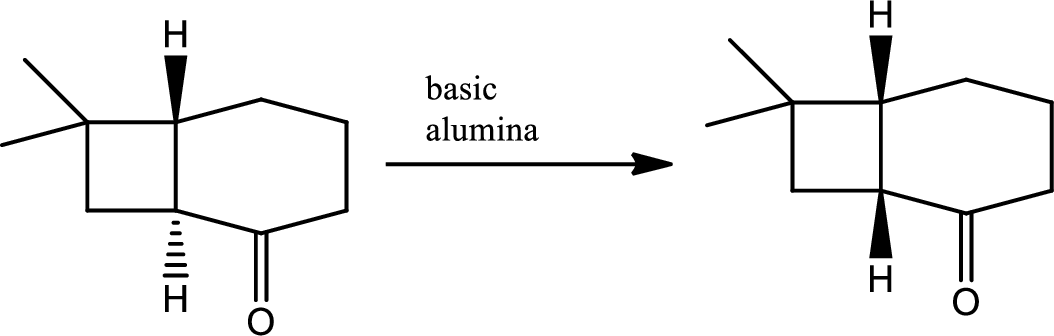
Concept introduction:
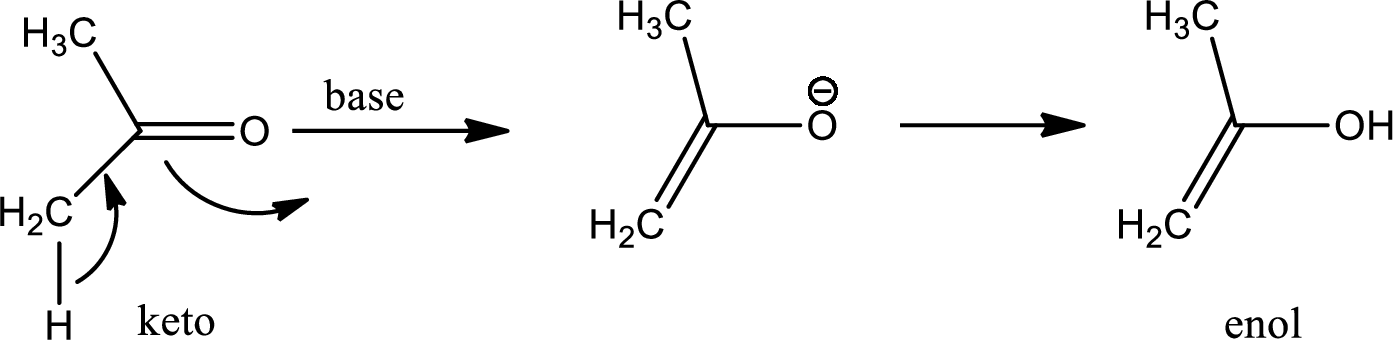
Tautomerisation:
Tautomers are structural isomers of a compound that readily interconverts into each other. The process of interconversion is called the tautomerisation and it involves simple proton transfer in intramolecular fashion.
One of the major examples of tautomerisation is keto–enol tautomerism. The mechanism is as follows,
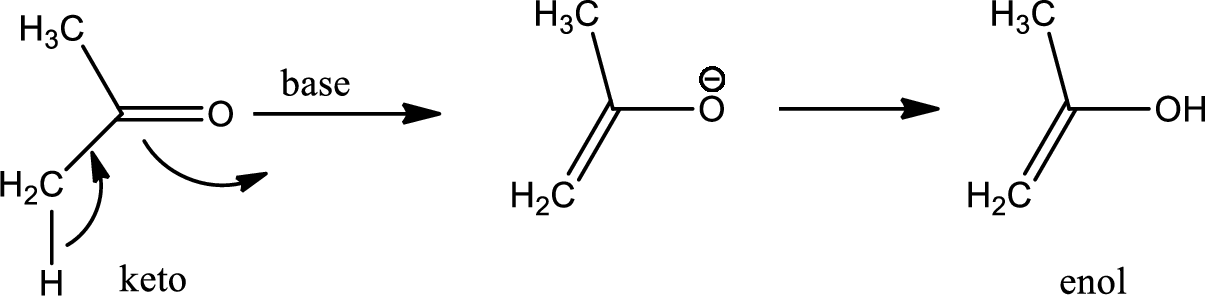
Among these keto and enol keto is more stable due to high bond energy of carbon oxygen double bond (
Explanation of Solution
Here basic alumina acts as base and abstract the enolisable hydrogen from left side because thus the enol form is much more stabilised due to formation of more substituted alkene.
But again the enol form tautomarises to keto form as the bond energy of carbon double bonded oxygen is more than single bonded carbon and oxygen. Thus the proton undergoes backside attack and so this product is formed.
The mechanism is as follows,
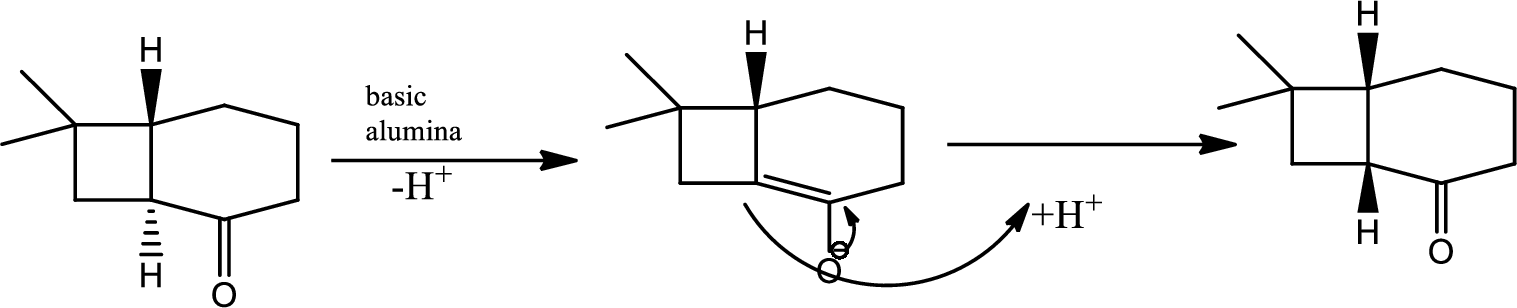
(c)
Interpretation:
The mechanism for the given step has to be given.
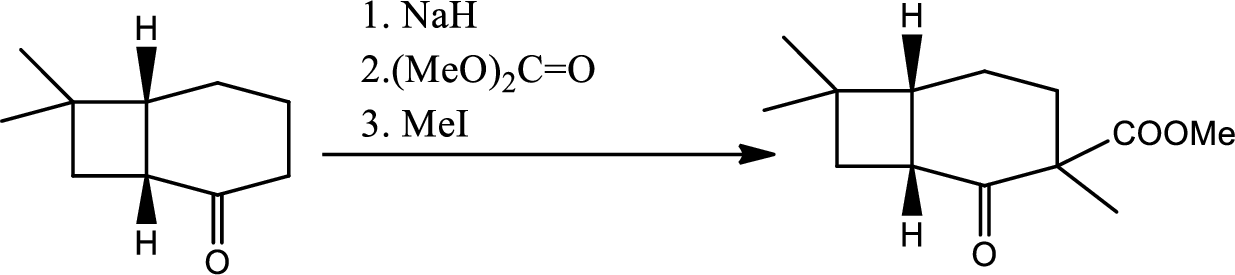
Concept introduction:
Acid-base reaction:
The species that donates proton or accepts lone pair of electrons are called acids and those who accepts proton or donates lone pair of electrons are called base.
The
Explanation of Solution
Here the reaction undergoes acid base reaction followed by
Here
Then again this leaving group
The mechanism is as follows,
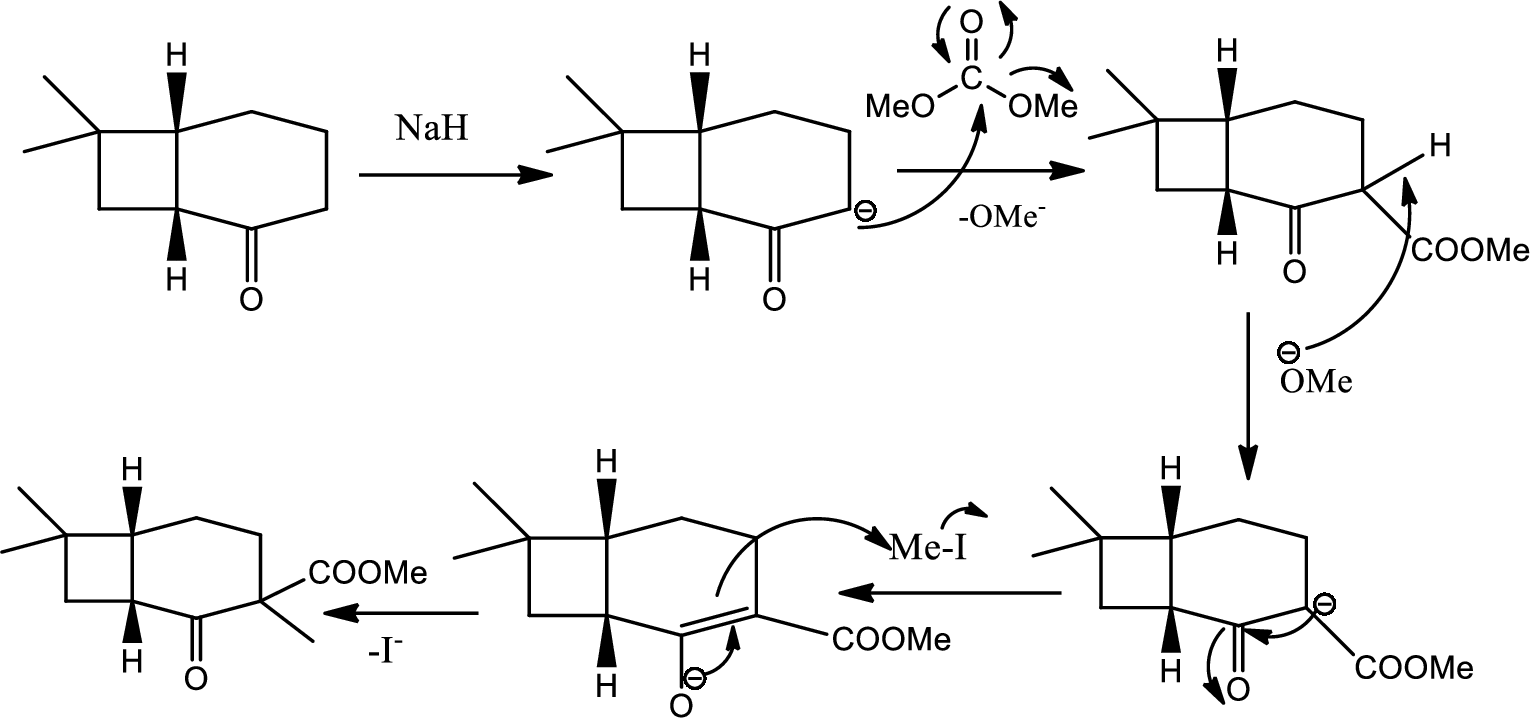
(d)
Interpretation:
Whether the stereochemistry of the added carbomethoxy group matters or not that has to be determined.
Concept introduction:
Steric hindrance:
Steric effects are non bondind interactions that influence the shape (conformation) and reactivity of the ions and molecules. Steric effects complement electronic effects which usually dictate shape and reactivity. Steric effects result from repulsive forces between overlapping electron clouds.
Steric hindrance is the consequence of steric effects. Steric hindrance is the slowing of chemical reactions due to steric bulk.
Explanation of Solution
From the later steps included in the reaction it can be concluded that the added carbomethoxy group’s stereochemistry matters as it is a bulky group and later on the reaction more bulkier rings are formed so it has to be properly oriented to the opposite side to avoid the steric hindrance.
(e)
Interpretation:
The structure of the compound A has to be given for the given reaction,
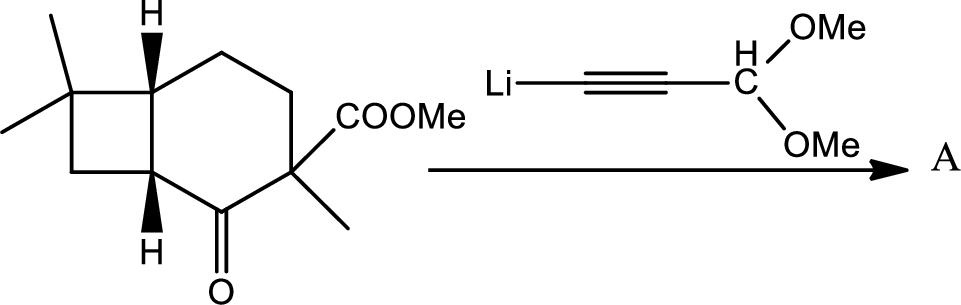
Concept introduction:
Nucleophilic addition reaction:
Nucleophilic addition reaction can be described as the addition reaction between the chemical compound that is electron deficient and having electron deficeint pi bonds with electron rich chemical compound known as nucleophile that results into disapperance of the pi bond amnd formation of new sigma bonds.
Addition of nucleophile to carbon heteroatom double or triple bonds shows variety as these bonds are polar due to high electrnegetive differences and hence the carbon atom carries a partial positive cahrge thus the carbon atom becomes the electrophilic centre and becomes primary target for nucleophile and thus the reactions occurs is aclled 1,2-nucleophilic addition reaction. The mechanism is as follows,
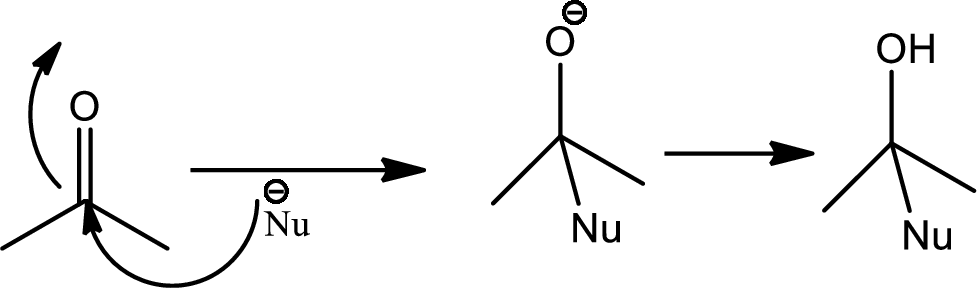
Organolithium compounds:
Organo lithium compounds are organometallic compounds containing carbon lithium bond. These act as nucleophile and bases also because of the high electronegetive difference between carbon and lithium and the bond becomes polar. Nucleophilic organolithium reagents can add to electrophilic carbonyl double bonds to form carbon-carbon bonds. They react with ketone or aldehyde to give alcohol i.e. they mainly undergoes 1,2-nucleophilic addition reaction
Explanation of Solution
Here the Organolithium compound is used as nucleophile which will attack the carbonyl carbon that behaves as electrophile and reduce it to alcohol. The oxygen lithium bond formed is more polar. The reaction is as follows,
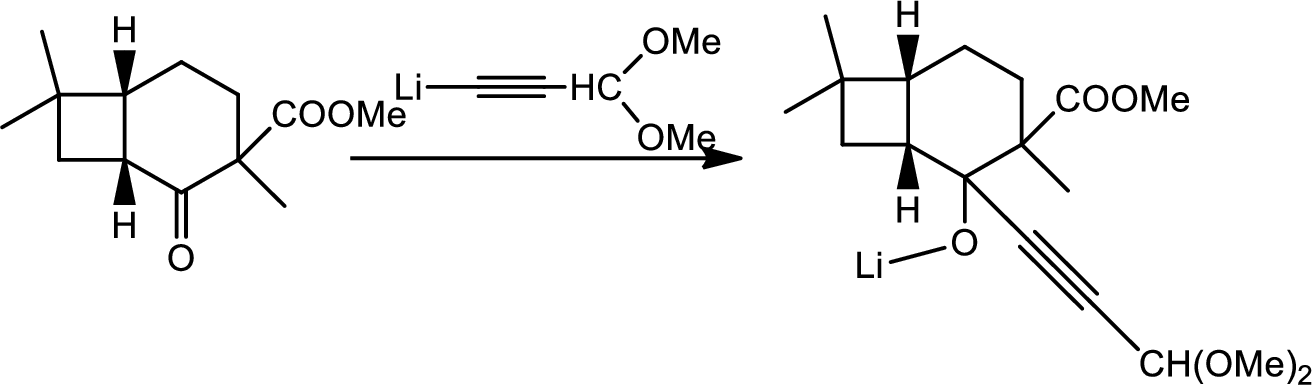
Thus the structure of compound A is,
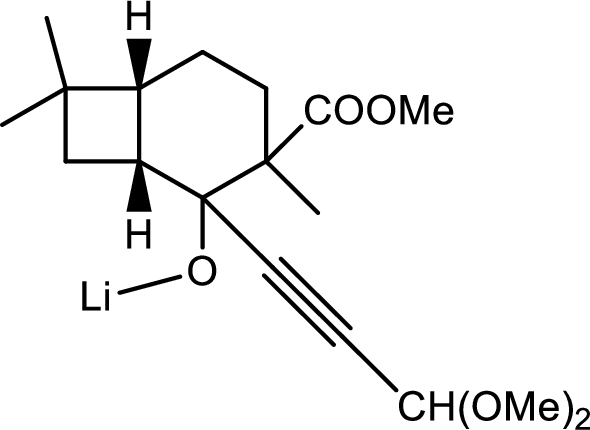
(f)
Interpretation:
The mechanism for the cyclised product given the question has to be given.
Concept introduction:
Reduction of alkyne:
Treatment of an alkyne with
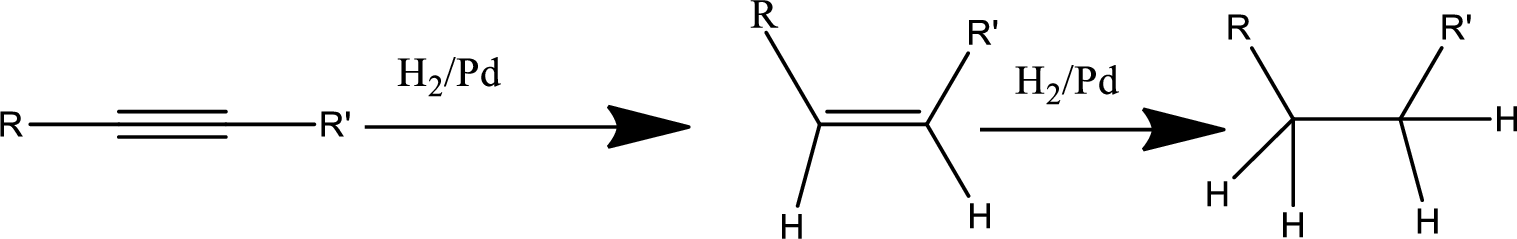
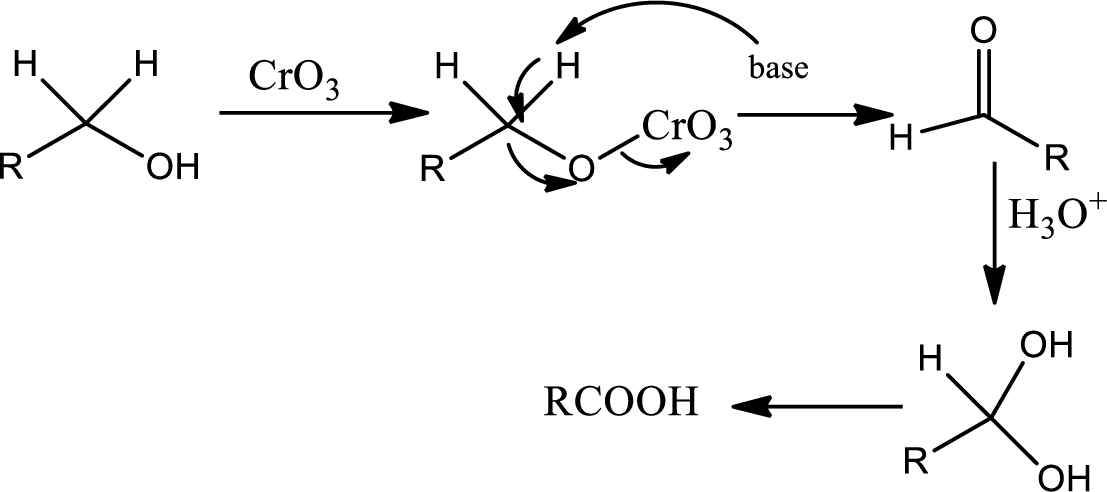
Oxidation by
Chromic acid is used as oxidising agent. It oxidises primary alcohol to acid and it oxidides seconadary alcohol to ketone in presence of acid. The mechanism as follows,
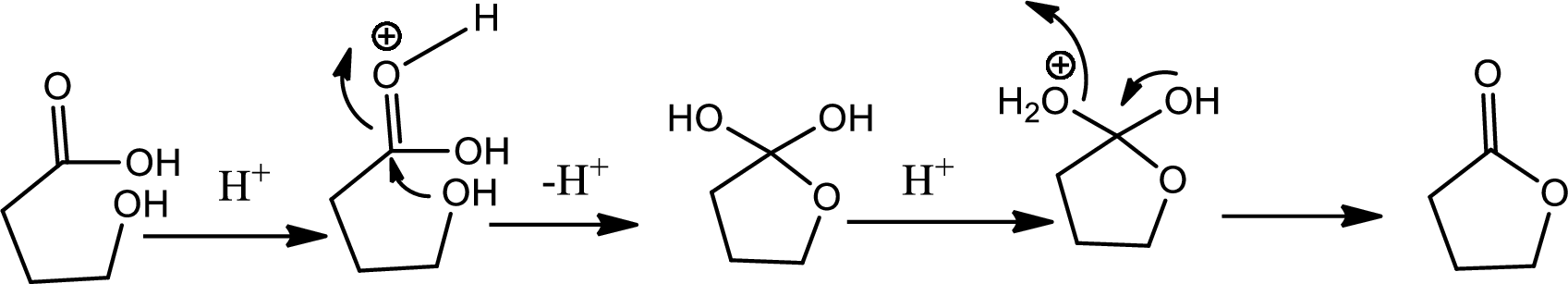
Lactone formation:
Lactones are cyclic carboxylic esters that are formed by intramolecular esterification of the corresponding hydroxycarboxylic acids which takes place spontaneously when th ering that is formed is five or six membered. The reaction pathway is as follows,
Explanation of Solution
In this pathway of lactone formation 1st the alkyne has been reduced to alkane… the reaction mechanism as follows,
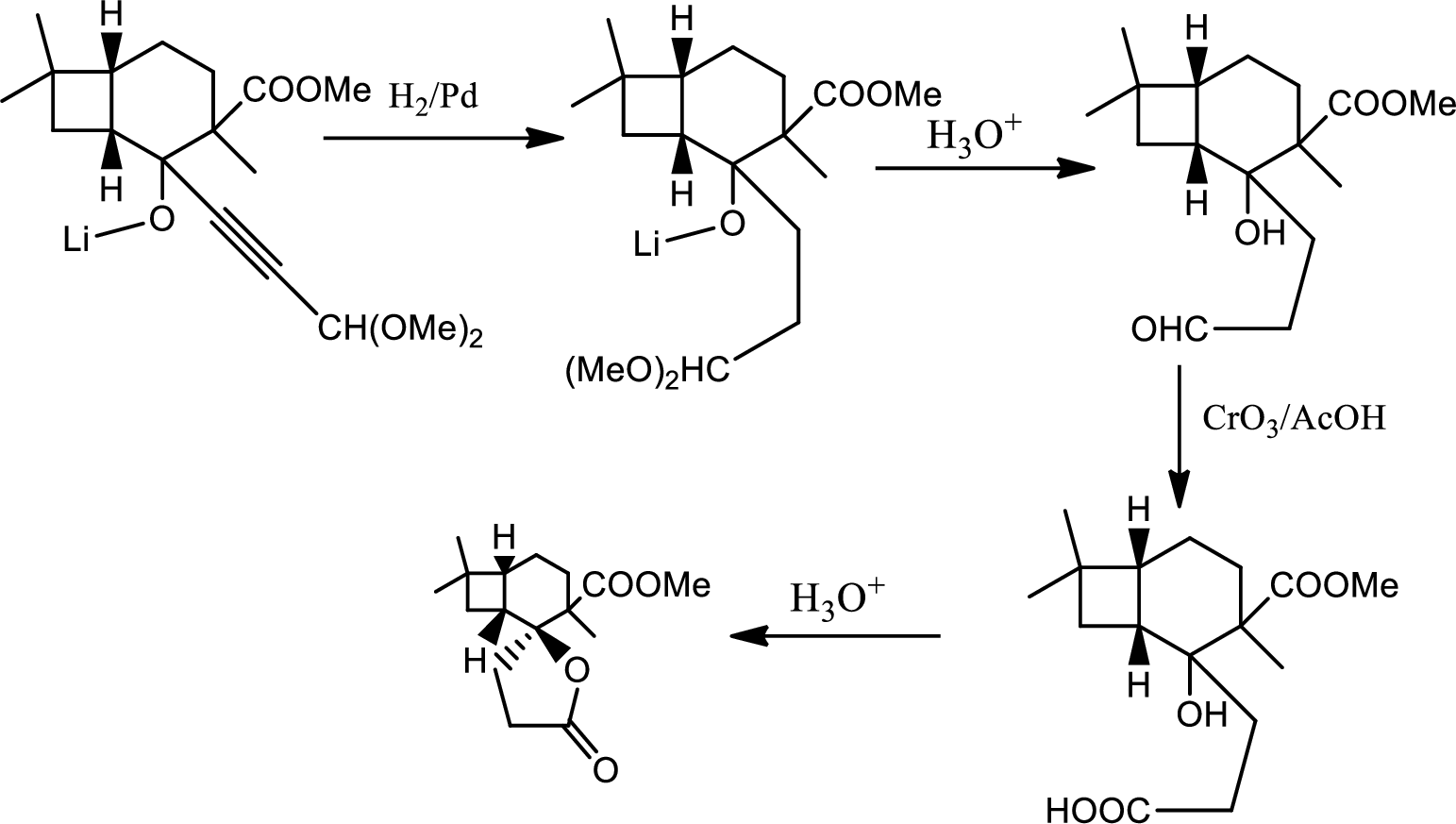
In the 2nd step the orthoester is oxidised to aldehyde which is further oxidised to acid with reaction with
(g)
Interpretation:
Mechanism for the given step has to be given,
Concept introduction:
Acid-base reaction:
The species that donates proton or accepts lone pair of electrons are called acids and those who accepts proton or donates lone pair of electrons are called base.
Explanation of Solution
The mechanism follows as,
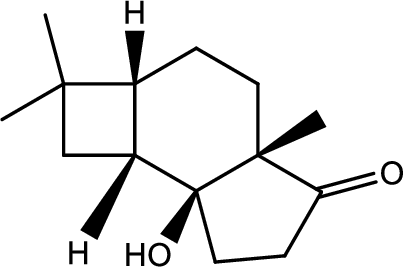
(h)
Interpretation:
Structure of product B has to be given.
Concept introduction:
Decarboxylation reaction:
This is the reaction where carboxyl acid group is eliminated along with carbon dioxide.
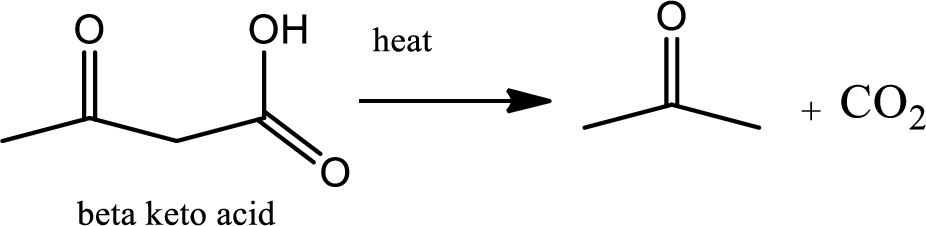
Explanation of Solution
The reaction pathway follows as,
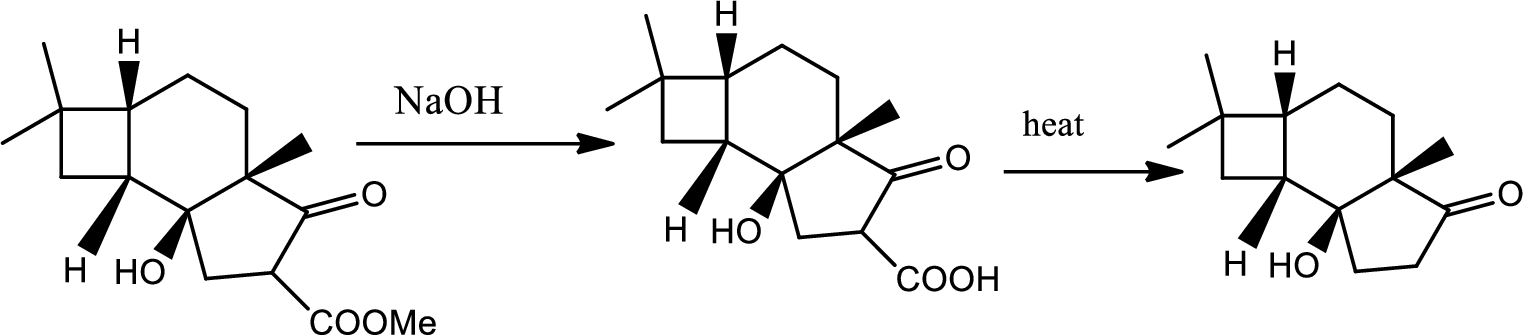
1st the ester is hydrolysed to acid followed by decarboxylation.
Hence the product B is,
(i)
Interpretation:
The reactions of B for given condition has to be shown.
Concept introduction:
Reduction of ketone:
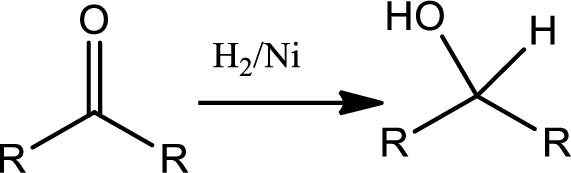
The
Explanation of Solution
The reaction goes as,
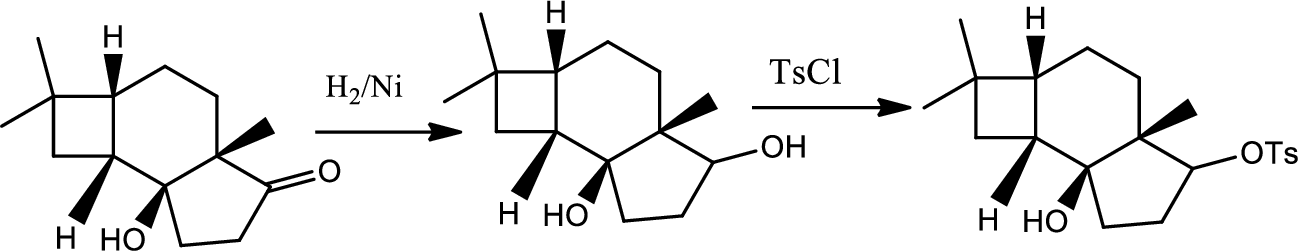
1st the ketone is reduced to alcohol which further undergoes
(j)
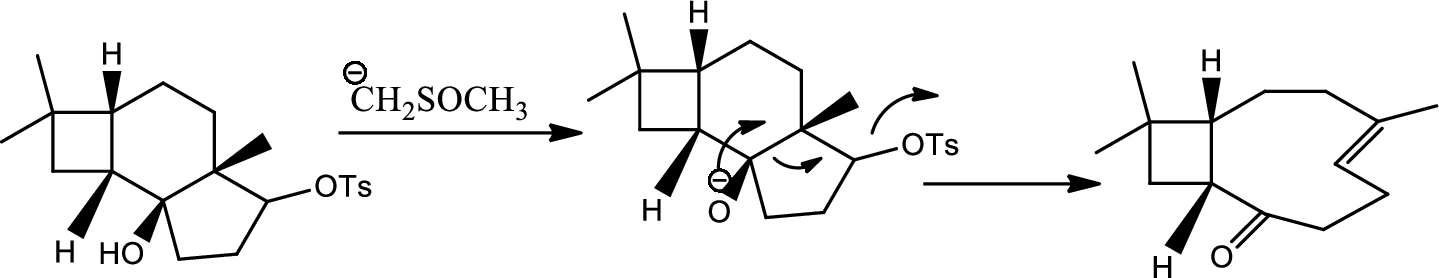
Interpretation:
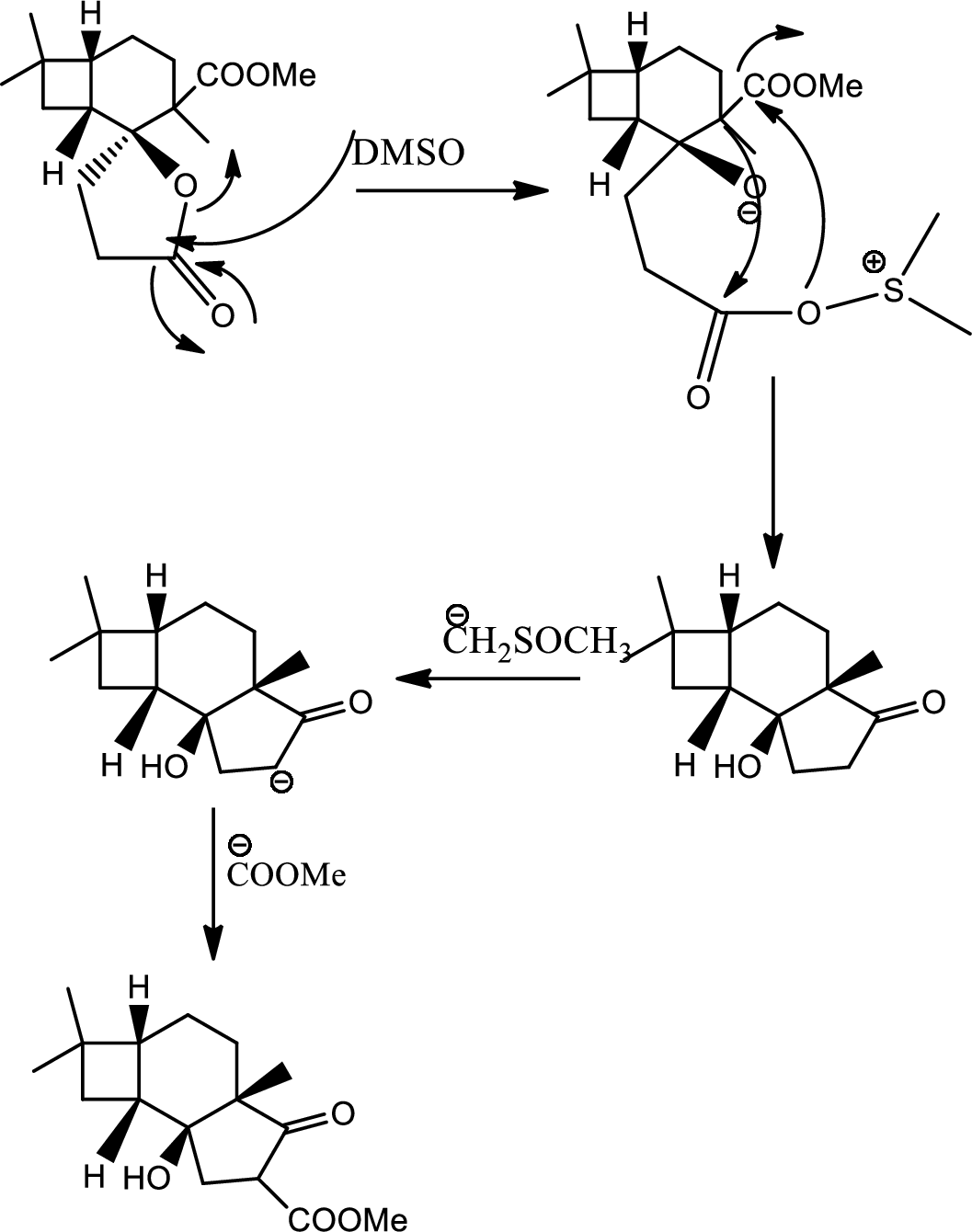
Mechanism of the ring opening reaction has to be shown.
Concept introduction:
Acid-base reaction:
The species that donates proton or accepts lone pair of electrons are called acids and those who accepts proton or donates lone pair of electrons are called base.
Explanation of Solution
DMSO here acts as a strong base and takes up the acidic hydrogen from alcohol. Thus to form strong carbon oxygen double bond the ring opens and
(k)
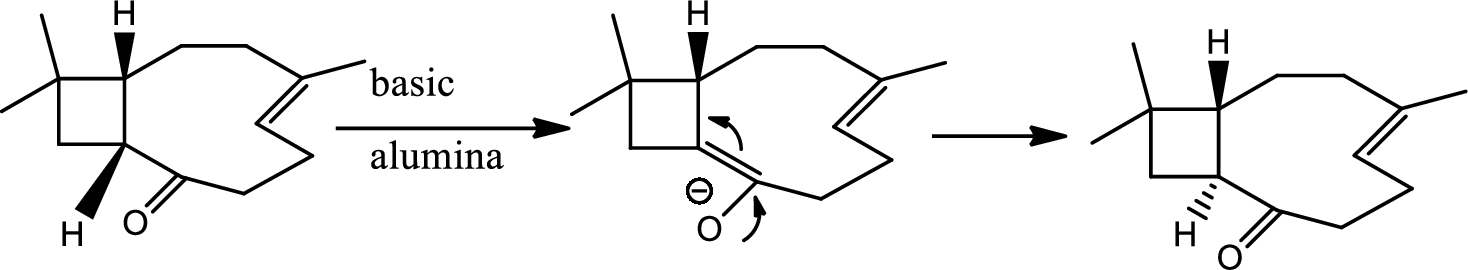
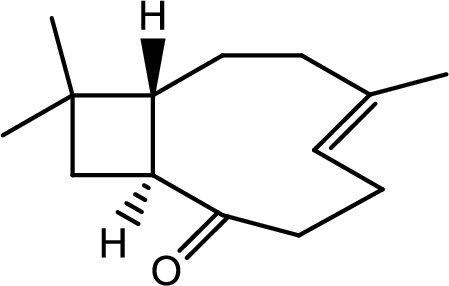
Interpretation:
The structure of C has to be determined.
Concept introduction:
Tautomerisation:
Tautomers are structural isomers of a compound that readily interconverts into each other. The process of interconversion is called the tautomerisation and it involves simple proton transfer in intramolecular fashion.
One of the major examples of tautomerisation is keto–enol tautomerism. The mechanism is as follows,
Among these keto and enol keto is more stable due to high bond energy of carbon oxygen double bond (
Explanation of Solution
Here basic alumina acts as base and abstract the enolisable hydrogen from left side because thus the enol form is much more stabilised due to formation of more substituted alkene.
But again the enol form tautomarises to keto form as the bond energy of carbon double bonded oxygen is more than single bonded carbon and oxygen. Thus the proton undergoes backside attack and so this product is formed.
The mechanism is as follows,
Hence the product C is,
(l)
Interpretation:
The reagents required for the transition given in the question has to be determined.
Concept introduction:
Wittig reaction:
Wittig reaction is the reaction of an aldehyde or ketone with a triphenyl phosphonium ylide to give an alkene and triphenylphosphine oxide. This is a step up reaction.
Explanation of Solution
The reaction as follows,
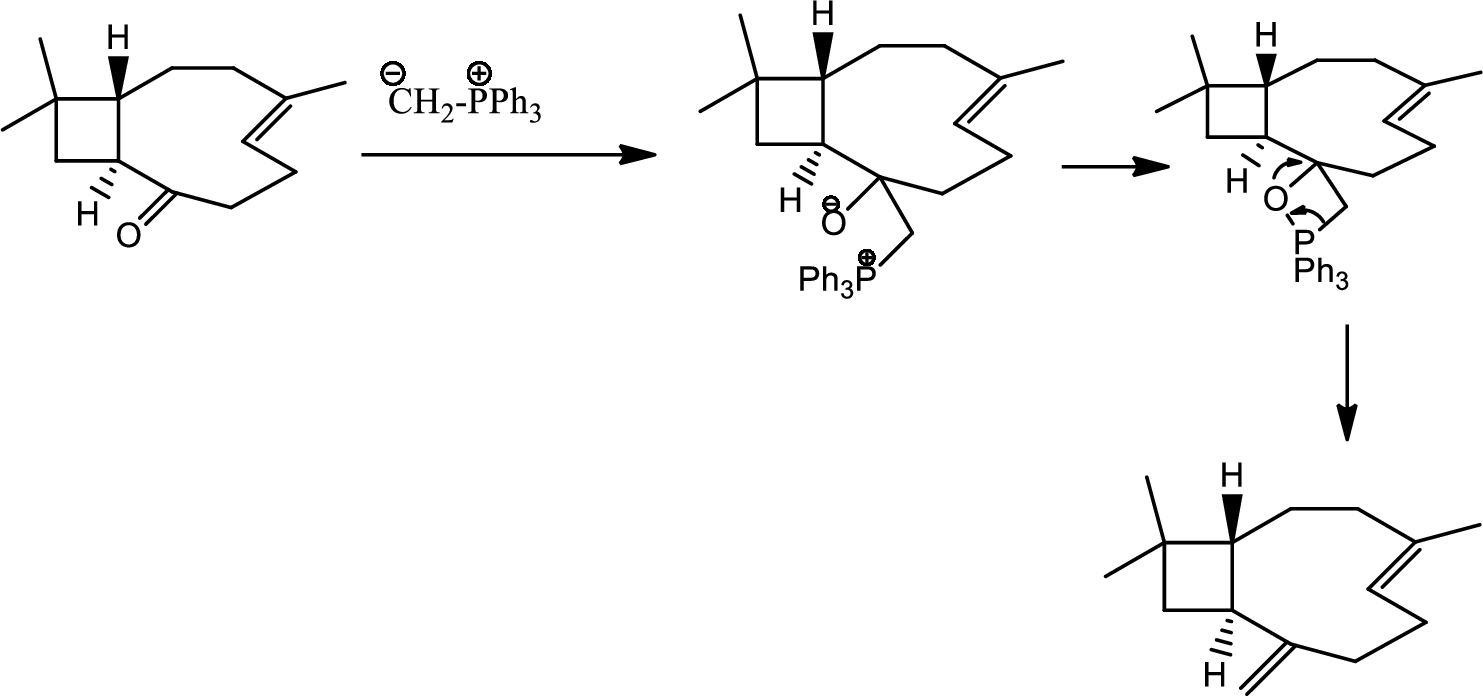
Thus the final product is obtained.
Want to see more full solutions like this?
Chapter 24 Solutions
Organic Chemistry
- What product would you expect to obtain from addition of Cl2 to 1,2-dimethylcyclohexene? Show the stereochemistry of the product.arrow_forwardCan you please check my work on the following ochem reaction scheme and let me know if it is correct or what is wrong... the question was: Consider 3,4-dimethylpiperidine being subjected to the following: Step 1: CH3I (excess); Step 2: NaOH, heat Step 3: CH3I (excess); Step 4: NaOH, heat Provide the bond line structures for the major organic product obtained in each step and discuss the regiochemistry for Step 2.arrow_forwardWhen Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% ofproduct A and 40% of product B. When the same reaction takes place at 60 °C, theproduct ratio is 10% A and 90% B.If you had a solution of pure A, and its temperature were raised to 60 °C, what wouldyou expect to happen? Propose a mechanism to support your prediction.arrow_forward
- Benzene, C6H6 undergoes substitution reaction with concentrated nitricacid, HNO3 to produce compound L. The reaction of compound L withbromine, Br2 in the presence of iron tribromide, FeBr3 produced compoundM. Benzene also undergoes Fridel-crafts alkylation reaction withchloroethane, CH3CH2Cl using catalyst N to produce compound P (i) Draw the structural formula of L, M and P (ii) State catalyst N. (iii) Show the formation of electrophile that will be reacted with benzene for theformation of compound P.arrow_forwardUemura and coworkers studied a time dependent Diels-Alder reaction which first formed the endo product as the major organic product and with time produced the exo product (J. Org. Chem. 2018, 83, 9300−9304). Show the endo and exo product for the reaction below. Which is the thermodynamic product and which is the kinetic product? Explain your reasoning.arrow_forwardSuppose you were told that each reaction is a substitution reaction, but you were not told the mechanism. Describe how you could conclude from the structure of the haloalkane or cycloalkene, the nucleophile, and the solvent that each reaction is an SN1 reaction.arrow_forward
- When anthracene is added to the reaction of chlorobenzene with concentrated NaOH at 350 °C, an interesting Diels–Alderadduct of formula C20H14 results. The proton NMR spectrum of the product shows a singlet of area 2 around d 3 and abroad singlet of area 12 around d 7. Propose a structure for the product, and explain why one of the aromatic rings ofanthracene reacted as a dienearrow_forwardExplain the following result. Although alkenes are generally more reactive than alkynes towards electrophiles, the reaction of Cl2 with but-2-yne can be stopped after one equivalent of Cl2 has been added.arrow_forwardClassify the carbocations given below as 1 °, 2 or 3 °, and write the product that will form the SN2 reaction on the most efficient substrate in a basic aqueous environment (H2O / HO).arrow_forward
- Show stereo chemistry and possible outcomes and the steps for the reaction.arrow_forwardAn unknown compound is treated with HBr in the dark. The product of this reaction is reacted with potassium t-butoxide. The product of reaction 2 is treated with HBr and peroxide. In reaction 4 the product of reaction 3 is treated with lithium acetylenide (C2H-). The product of reaction4 is reduced with hydrogen using platinum as a catalyst. This final product was determined to be n-pentylcyclopentane. What is the name of the original unknown compound?arrow_forwardExplain the following result. Although alkenes are generally more reactive than alkynes toward electrophiles, the reaction of Cl2 with but-2-yne can be stopped after one equivalent of Cl2 has been added.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

