
(a)
Interpretation: The primary, secondary or tertiary amine in the given compound is to be classified.
Concept introduction:
In secondary amines, two of the hydrogen atoms of ammonia are replaced by alkyl or aryl group.
In tertiary amines, all hydrogen atoms of ammonia are replaced by alkyl or aryl group.
Answer to Problem 25.1P
Each amine in the given compound is classified as,
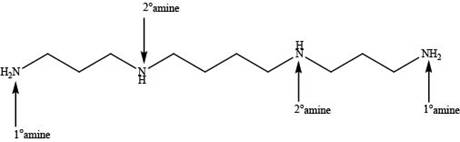
Explanation of Solution
The given compound is shown below.
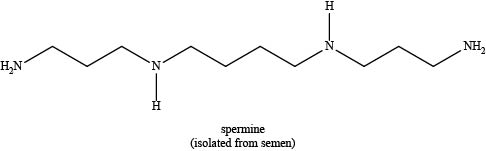
Figure 1
In spermine, four nitrogen atoms are present. Among these four nitrogen atoms, two are primary amines that contain one alkyl group on nitrogen atom and two are secondary amines that contain two alkyl groups on nitrogen atom as shown below.
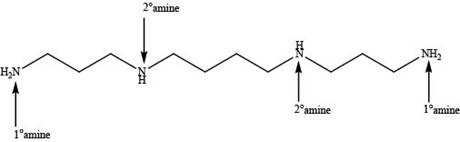
Figure 2
In spermine, primary and secondary amines are present as shown in Figure 2.
(b)
Interpretation: The primary, secondary or tertiary amine in the given compound is to be classified.
Concept introduction: Amines are nitrogen-containing organic compounds. The general formula of amines is
In secondary amines, two of the hydrogen atoms of ammonia are replaced by alkyl or aryl group.
In tertiary amines, all hydrogen atoms of ammonia are replaced by alkyl or aryl group.
Answer to Problem 25.1P
Each amine in the given compound is classified as,
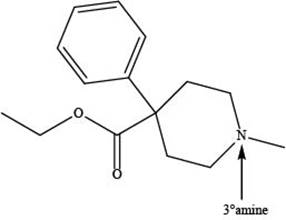
Explanation of Solution
The given compound is shown below.
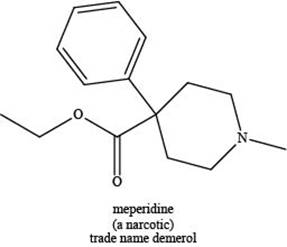
Figure 3
Meperidine contains one nitrogen atom bonded to three alkyl groups. Hence, it is a tertiary amine as shown below.

Figure 4
In meperidine, tertiary amine is present as shown in Figure 4.
Want to see more full solutions like this?
Chapter 25 Solutions
Organic Chemistry -Study Guide / Solution Manual (Custom)
- Which type of amine is phentermine? a) a primary aliphatic amine b) a primary aromatic amine c) a tertiary aliphatic amine d) a tertiary aromatic aminearrow_forward1. What are the hazards and benefits of Amines? 2. What are the hazards and benefits of Nitriles? 3. What are the hazards and benefits of Nitro Compounds?arrow_forwardWhich type of amine is (s)-methamphetamine? a) a primary aliphatic amine b) a primary aromatic amine c) a secondary aliphatic amine d) a secondary aromatic aminearrow_forward
- N-Methylpyrrolidine has a boiling point of 81 °C, and piperidine has a boiling point of 106 °C. Tetrahydropyran has a boiling point of 88 °C, and cyclopentanone has a boiling point of 141 °C. These two isomershave a boiling point difference of 53 °C. Explain why the two oxygen-containing isomers have a much larger boilingpoint difference than the two amine isomersarrow_forwardMatch the description to one of the compounds E– H. a. a compound that contains a 1 ° amine and a 1 ° amide b. a compound that contains a 1 ° amine and a 2 ° amide c. a compound that contains a 2 ° amine and a 3 ° amide d. a compound that contains a 3 ° amine and a 3 ° amidearrow_forwardWhat nitro compound, nitrile, and amide are reduced to each compound?arrow_forward
- Aromatic amines are _________ neutral because of the polarity are unique as they do not behave like any of the other amines acidic, but stronger than the aliphatic amines basic, but weaker than aliphatic aminesarrow_forward1. Which statement best described the ability of amines to hydrogen bond? A. Primary, secondary and tertiary amines can all hydrogen bond with molecules identical to themselves. B. Primary and secondary amines can hydrogen bond with molecules identical to themselves, but tertiary amines cannot. C. Only primary amines can hydrogen bond with molecules identical to themselves. Secondary and tertiary amines cannot. D. Primary, secondary and tertiary amines cannot hydrogen bond with molecules identical to themselves, but the can hydrogen bond with water.arrow_forwardA) Name the following amine. H3C−CH2−CH2−NH−CH2−CH2−CH3 Spell out the full name of the compound. B ) Name the following amine. CH3−CH2−NH−CH2−CH2−CH3 Spell out the full name of the compound.arrow_forward
- amines Name and classify each compoundarrow_forwardArrange the following amines in order of increasing boiling point, lowest bp to highest bp:(CH3)2CHCH2CH2NH2, (CH3)2CHN(CH3)2, and (CH3)2CHCH2NHCH3.arrow_forwardMany drugs are sold as their hydrochloride salts (R2NH2 + Cl−), formed by reaction of an amine (R2NH) with HCl . Question: Draw the product (a hydrochloride salt) formed by reaction ofacebutolol with HCl. Acebutolol is a β blocker used to treat high bloodpressure.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning





