
Concept explainers
a)
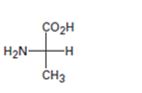
Interpretation:
The Fischer projection formula is to be converted in to a tetrahedral representation and its configuration as R or S is to be assigned.
Concept introduction:
In Fischer projection formula, a tetrahedral carbon is represented by two crossed lines. The horizontal line represents bonds coming out of the page and vertical lines represent bonds moving in to the page.
For assigning R or S configuration, the four groups attached to the chiral center are arranged in the order of priority by applying sequence rules. The molecule is then oriented in such a way that the group of lowest priority points away from the viewer. If the arrangement of highest priority to second highest priority to third highest priority is clockwise then R configuration is assigned. If the arrangement of highest priority to second highest priority to third highest priority is counterclockwise then S configuration is assigned.
To covert:
The Fischer projection formula in to a tetrahedral representation and to assign its configuration as R or S.
b)
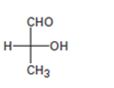
Interpretation:
The Fischer projection formula is to be converted in to a tetrahedral representation and its configuration as R or S is to be assigned.
Concept introduction:
In Fischer projection formula, a tetrahedral carbon is represented by two crossed lines. The horizontal line represents bonds coming out of the page and vertical lines represent bonds moving in to the page.
For assigning R or S configuration, the four groups attached to the chiral center are arranged in the order of priority by applying sequence rules. The molecule is then oriented in such a way that the group of lowest priority points away from the viewer. If the arrangement of highest priority to second highest priority to third highest priority is clockwise then R configuration is assigned. If the arrangement of highest priority to second highest priority to third highest priority is counterclockwise then S configuration is assigned.
To covert:
The Fischer projection formula in to a tetrahedral representation and to assign its configuration as R or S.
c)
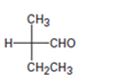
Interpretation:
The Fischer projection formula is to be converted in to a tetrahedral representation and its configuration as R or S is to be assigned.
Concept introduction:
In Fischer projection formula, a tetrahedral carbon is represented by two crossed lines. The horizontal line represents bonds coming out of the page and vertical lines represent bonds moving in to the page.
For assigning R or S configuration, the four groups attached to the chiral center are arranged in the order of priority by applying sequence rules. The molecule is then oriented in such a way that the group of lowest priority points away from the viewer. If the arrangement of highest priority to second highest priority to third highest priority is clockwise then R configuration is assigned. If the arrangement of highest priority to second highest priority to third highest priority is counterclockwise then S configuration is assigned.
To covert:
The Fischer projection formula in to a tetrahedral representation and to assign its configuration as R or S.
Trending nowThis is a popular solution!

Chapter 25 Solutions
Organic Chemistry
- 1. What is the relationship between the following two compounds? a.stereoisomersb.identicalc.constitutional isomers 2. Assign the absolute configuration of the chirality center as R or S.arrow_forward1. Draw all of the possible stereoisomers of the compound shown. Group them asenantiomeric pairs, or indicate any meso compounds.2. Label all stereocenters with the absolute stereochemistry (R or S).arrow_forwardDraw the structure of Ethyl 2-deoxoy-2-fluoro-a-D-galactopyranoside in: i) cyclized Fischer projection ii) in its most stable chair configuration iii) in its least stable chair confirmationarrow_forward
- As a fischer projection is the compound R or S and which group is the highest priority ?arrow_forwardAssign R or S configurations to each stereogenic center of isoborneol and borneol. Give the relationship between these two stereoisomers.arrow_forwardConvert the following compound to a Fischer projection and label each stereogenic center as R or S by providing the missing information in the labeled Fischer projection provided below. Give the configurations of A, B and C and the substituents from 1 to 8.arrow_forward
- Find a constitutional isomer of C7H16 that has only one tetrahedral stereocenter and then make a Fischer projection of the R-stereoisomer, placing the group with priority 4 on top and priority 2 on bottom.arrow_forwardAssign R/S to each of the chirality centers shown below.arrow_forwardConsider a mixture that is 60% (+) enantiomer and 40% (-) enantiomer. In which direction will the mixture rotate plane-polarized light? What is the enantiomeric excess of the mixture?arrow_forward
- Draw the structure for a compound with molecular formula C2H2I2F2 a. that is optically inactive because it does not have an asymmetric center. b. that is optically inactive because it is a meso compound. c. that is optically active.arrow_forwardDraw all of the possible stereoisomers of the compound shown. Group them as enantiomeric pairs, or indicate any meso compounds. Label all stereocenters with the absolute stereochemistry (R or S).arrow_forwardSalinosporamide A, isolated from a microbe called Salinispora tropica has shown promise as an inhibitor of cancer cell growth. Select the chiral centers in the following structure; the selected centers will appear highlighted green.arrow_forward
