
Concept explainers
a)
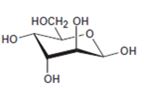
Interpretation:
The open-chain form of the sugar given is to be drawn.
Concept introduction:
The pyranose form is a cyclic hemiacetal form with a six membered ring formed by the nucleophilic addition of the –OH group on C5 to the C1 carbonyl group. The furanose form is a cyclic hemiacetal form with a five membered ring formed by the nucleophilic addition of the –OH group on C5 to the C2 carbonyl group.
The orientation of –OH group differs in α- and β- anomers. In α- anomer the OH on C1 is cis to the –OH at the lowest chirality center in Fischer projection while in β- anomer the –OH on C1 is trans to the –OH at the lowest chirality center in Fischer projection.
D sugars have the –O- at C5 on the right in the uncoiled form while L sugars have -O- at C5 on the left.
To draw:
The open-chain form of the sugar given.
b)
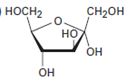
Interpretation:
The open-chain form of the sugar given is to be drawn.
Concept introduction:
The pyranose form is a cyclic hemiacetal form with a six membered ring formed by the nucleophilic addition of the –OH group on C5 to the C1 carbonyl group. The furanose form is a cyclic hemiacetal form with a five membered ring formed by the nucleophilic addition of the –OH group on C5 to the C2 carbonyl group.
The orientation of –OH group differs in α- and β- anomers. In the α- anomer the OH on C1 is cis to the –OH at the lowest chirality center in Fischer projection, while in β- anomer the –OH on C1 is trans to the –OH at the lowest chirality center in Fischer projection.
D sugars have the –O- at C5 on the right in the uncoiled form while L sugars have -O- at C5 on the left.
To draw:
The open-chain form of the sugar given.
c)
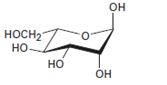
Interpretation:
The open-chain form of the sugar given is to be drawn.
Concept introduction:
The pyranose form is a cyclic hemiacetal form with a six membered ring formed by the nucleophilic addition of the –OH group on C5 to the C1 carbonyl group. The furanose form is a cyclic hemiacetal form with a five membered ring formed by the nucleophilic addition of the –OH group on C5 to the C2 carbonyl group.
The orientation of –OH group differs in α- and β- anomers. In the α- anomer the OH on C1 is cis to the –OH at the lowest chirality center in Fischer projection, while in β- anomer the –OH on C1 is trans to the –OH at the lowest chirality center in Fischer projection.
D sugars have the –O- at C5 on the right in the uncoiled form while L sugars have -O- at C5 on the left.
To draw:
The open-chain form of the sugar given.
Trending nowThis is a popular solution!

Chapter 25 Solutions
Organic Chemistry
- which of the following D-aldopentoses could be possible structures for xarrow_forwardIf you take crystals of the disaccharide lactose the natural substrate for beta-galactosidase and drop them in water, why doesn’t the lactose immediately react with the water and come apart into glucose and galactose?arrow_forwardDraw the structure of maltose and cellubiose. Mark the difference in both structure.arrow_forward
- Draw the structures of an aldopentose and a ketohexose.arrow_forwardWhat is essentially the difference between a-glucose and P-glucose? What is meant by pyranose structure of glucose?arrow_forwardQ1: What structural relationship is indicated by the term D-sugar? Why are (+) glucose and (−) fructose both classified as D-sugars? Write the principal functional roles of D-glucose, D-fructose and galactosearrow_forward

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,





