
Concept explainers
Inhibition of Purine and Pyrimidine
(a)
Interpretation:
A reaction of purine or pyrimidine metabolism is affected by the azaserine.
Concept Introduction:
Azaserine and DON are equivalents of glutamine and bind to glutamine-binding proteins. They can react with nucleophiles, leading to covalent modification and inactivation.
Answer to Problem 4P
Azaserine and DON are equivalents of glutamine and bind to glutamine-binding proteins. They can react with nucleophiles, leading to covalent modification and inactivation. The following reactions in purine synthesis are sensitive to these inhibitors.
There is another reaction that's sensitive to those inhibitors for the synthesis of pyrimidines.
Explanation of Solution
There are 3 structures that are analogs to amino acid, and that they are azaserine (O-diazoacetvl-Lserine) and a connected compound DON, and 6-diaxo-5-oxo-L-norleucine. Their structures are shown below:
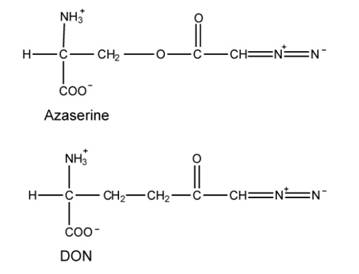
Azaserine and DON are equivalents of glutamine and bind to glutamine-binding proteins. They can react with nucleophiles, leading to covalent modification and inactivation. The following reactions in purine synthesis are sensitive to these inhibitors.
There is another reaction that's sensitive to those inhibitors for the synthesis of pyrimidines.
(b)
Interpretation:
A reaction of purine or pyrimidine metabolism is affected by the methotrexate.
Concept Introduction:
Methotrexate is analogous to dihydrofolate in this it competes with folic acid with high affinity for binding sites on enzymes. Some reactions in purine synthesis are affected.
Answer to Problem 4P
The formation of tTMP in pyrimidine synthesis will be blocked by methotrexate sodium. The reaction is seen below.
Explanation of Solution
Methotrexate is analogous to dihydrofolate in this it competes with folic acid with high affinity for binding sites on enzymes. Some reactions in purine synthesis are affected.
The formation of tTMP in pyrimidine synthesis will be blocked by methotrexate sodium. The reaction is seen below.
(c)
Interpretation:
A reaction of purine or pyrimidine metabolism is affected by the sulfonamides.
Concept Introduction:
Sulfonamides are structurally the same as para-aminobenzoic acid. They inhibit the assembly of folacin in bacterium.
Answer to Problem 4P
Ester synthesis in animals won't be littered with sulfonamides since folacin is an element of a dietary demand for animals.
Explanation of Solution
Sulfonamides are structurally the same as para-aminobenzoic acid. They inhibit the assembly of folacin in bacterium. This inhibition can happen as a result of they cannot produce folacin because of lack of substrate. Ester synthesis in animals won't be littered with sulfonamides since folacin is an element of a dietary demand for animals.
(d)
Interpretation:
A reaction of purine or pyrimidine metabolism is affected by the allupurinol.
Concept Introduction:
Xanthine enzyme can hydroxylate Zyloprim, a suicide substance of organic compound enzyme, to create alloxantine that binds to the catalyst and inactivates it.
Answer to Problem 4P
Organic compound is regenerate to uric acid via the catalyst organic compound enzyme, and excrement acids will not form as a result of its blocked because of each nucleoside and nucleoside that are metabolized to organic compound.
Explanation of Solution
Xanthine enzyme can hydroxylate Zyloprim, a suicide substance of organic compound enzyme, to create alloxantine that binds to the catalyst and inactivates it. Organic compound is regenerate to uric acid via the catalyst organic compound enzyme, and excrement acids will not form as a result of its blocked because of each nucleoside and nucleoside that are metabolized to organic compound.
(e)
Interpretation:
A reaction of purine or pyrimidine metabolism is affected by the 5-fluorouracil.
Concept Introduction:
5-fluorouracil is converted to 5-fluorodeoxyuridylate (FdUMP), which is a powerful inhibitor of thymidylate synthase.
Answer to Problem 4P
Even though 5-fluorouracil is not a significant inhibitor of nucleotide metabolism, it is converted to 5-fluorodeoxyuridylate (FdUMP), which is a powerful inhibitor of thymidylate synthase.
Explanation of Solution
Even though 5-fluorouracil is not a significant inhibitor of nucleotide metabolism, it is converted to 5-fluorodeoxyuridylate (FdUMP), which is a powerful inhibitor of thymidylate synthase.
Want to see more full solutions like this?
Chapter 26 Solutions
Biochemistry
- What is the biochemical rationale for the isomerization of glucose 6- phosphate to fructose 6-phosphate and its subsequent phosphorylation to form fructose 1,6-bisphosphate?arrow_forwardIs trimethoprim a mechanism-based inhibitor of bacterial dihydrofolate reductase?arrow_forwardWhat beneficial properties of rasagiline make it useful for the treatment of Alzheimer’s disease?arrow_forward
- Insulin-dependent diabetes is often accompanied by hypertriglyceridemia, which is an excess blood level of triacylglycerols in the form of very low density lipoproteins. Suggest a biochemical explanation.arrow_forwardDraw a schematic illustration of the hydrolysis of N-acetylphenylalaninamide catalyzed byalpha-chymotrypsin in which you indicate the important catalytic residues in the active site and how thesubstrate undergoes transformation to products through two different tetrahedral intermediates.arrow_forwardThe interconversion of DHAP and GAP greatly favors the formation of DHAP at equilibrium. Yet the conversion of DHAP by triose phosphate isomerase proceeds readily. Whyarrow_forward
- What is the biochemical rationale for the use of low dose aspirin in patients withcoronary artery disease? Why is Ibuprofen not a good substitute for aspirin in thiscase?arrow_forwardAn inhibitor that specifically labels chymotrypsin at histidine 57 is Ntosylamido-l-phenylethyl chloromethyl ketone. How would you modify the structure of this inhibitor to label the active site of trypsin?arrow_forwardWhat is bioavilability of dacarbazine?arrow_forward
- What is the biochemical basis of allopurinol treatment for gout?arrow_forwardAbout cardiotonic glycosides, based on a digoxin structure in the image: Classify the α,β-unsaturated lactone present in digoxin. What is the mechanism of the pharmacological action of digoxin, explain in detail?arrow_forwardWhat is the biochemical rationale for the use of creatine?arrow_forward
 BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
