
Concept explainers
(a)
Interpretation: The given reaction is to be classified as an electrocyclic reaction, cycloaddition, or a sigmatropic rearrangement. The sigma bonds that are broken or formed in the given reaction are to be labeled.
Concept introduction: A
Answer to Problem 27.1P
The reaction is classified as an electrocyclic reaction. The sigma bond broken or formed in the given reaction is,
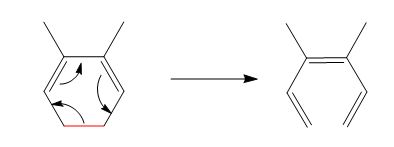
Explanation of Solution
The given reaction is shown below.

Figure 1
The sigma bond that is broken during the conversion of reactant into product is shown in red. The new bonds formed during the conversion are shown by curved arrows in the reactant. In the product, the ring is opened due to the rearrangement of
The reaction is classified as an electrocyclic reaction. The sigma bond that is broken or formed in the given reaction is labeled in Figure 1.
(b)
Interpretation: The given reaction is to be classified as an electrocyclic reaction, cycloaddition, or a sigmatropic rearrangement. The sigma bonds that are broken or formed in the given reaction are to be labeled.
Concept introduction: A chemical reaction that involves
Answer to Problem 27.1P
The reaction is classified as a cycloaddition reaction. The sigma bonds broken or formed in the given reaction are,
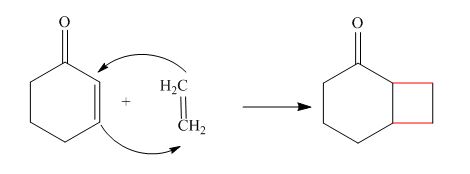
Explanation of Solution
The given reaction is shown below.

Figure 2
The sigma bonds that are formed during the conversion of reactant into a product are shown in red. In the reaction,
The reaction is classified as a cycloaddition reaction. The sigma bonds broken or formed in the given reaction are labeled in Figure 2.
(c)
Interpretation: The given reaction is to be classified as an electrocyclic reaction, cycloaddition, or a sigmatropic rearrangement. The sigma bonds that are broken or formed in the given reaction are to be labeled.
Concept introduction: A chemical reaction that involves
Answer to Problem 27.1P
The reaction is classified as an electrocyclic reaction. The sigma bond broken or formed in the given reaction is,
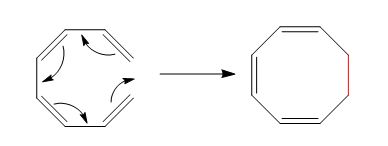
Explanation of Solution
The given reaction is shown below.

Figure 3
The sigma bond that is formed during the conversion of reactant into product is shown in red. The new bonds formed during the conversion are shown by curved arrows in the reactant. In the product, the ring gets closed due to the rearrangement of
The reaction is classified as an electrocyclic reaction. The sigma bond broken or formed in the given reaction is labeled in Figure 3.
(d)
Interpretation: The given reaction is to be classified as an electrocyclic reaction, cycloaddition, or a sigmatropic rearrangement. The sigma bonds that are broken or formed in the given reaction are to be labeled.
Concept introduction: A chemical reaction that involves
Answer to Problem 27.1P
The reaction is classified as a sigmatropic reaction. The sigma bonds broken or formed in the given reaction are,
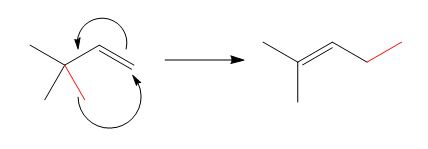
Explanation of Solution
The given reaction is shown below.

Figure 4
The sigma bond that is broken and formed during the conversion of reactant into product is shown in red. The product indicates that
The reaction is classified as a sigmatropic reaction. The sigma bonds broken or formed in the given reaction are labeled in Figure 4.
Want to see more full solutions like this?
Chapter 27 Solutions
ORGANIC CHEM W/CONNECT & MODEL KIT >CI
- What other alkene is also formed along with Y in Sample Problem 9.3? What alkenes would form from X if no carbocation rearrangement occurred?arrow_forwardWhat other alkene is also formed along with Y in Sample Problem 9.3 (Attached) ? What alkenes would form from X if no carbocation rearrangement occurred?arrow_forwardDoes cis- or trans-1-bromo-4-tert-butylcylohexane react faster in an E2reaction?arrow_forward
- Addition of HCl to alkene X forms two alkyl halides Y and Z. Explain why addition of HCl occurs at the indicated C=C (called anexocyclic double bond), rather than the other C=C (called anendocyclic double bond).arrow_forwardAlkene A can be isomerized to isocomene, a natural product isolated from goldenrod, by treatment with TsOH. Draw a stepwise mechanism for this conversion. (Hint: Look for a carbocation rearrangement.)arrow_forwardWhich reactions are stereoselective?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
