
(a)
Interpretation: The product of given reaction has to be drawn.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In an electrocyclic reaction “one new sigma- bond is formed or brocken.”
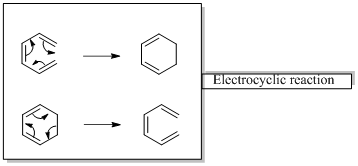
Woodward –Hoffmann rules are the set of rules used to vindicate or predict certain aspects of the stereo chemical outcome and activation energy of pericyclic reactions.
Woodward – Hoffmann rules for Electrocyclic reactions are listed below
A photochemical reaction takes place when a reactant absorbs light and a thermal reaction takes place without the absorption of light.
Woodward – Hoffmann rules for the configuration of electrocyclic reactions are,
(b)
Interpretation: The product of given reaction has to be drawn.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In an electrocyclic reaction “one new sigma- bond is formed or brocken.”

Woodward –Hoffmann rules are the set of rules used to vindicate or predict certain aspects of the stereo chemical outcome and activation energy of pericyclic reactions.
Woodward – Hoffmann rules for Electrocyclic reactions are listed below
A photochemical reaction takes place when a reactant absorbs light and a thermal reaction takes place without the absorption of light.
Woodward – Hoffmann rules for the configuration of electrocyclic reactions are,
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
- Items 49-51: Given the following reaction: CH3 H. CH3 49. What is the product of the reaction? A. В. C. D. 50. Which MO will be involved in this electrocyclic reaction? 888888 888888 888888 A. B. C. 51. The reaction will go through: A. a conrotatory motion B. a disrotatory motion C. none of thesearrow_forward13. Which of the following statements is INCORRECT? A. Radicals are very reactive species. B. Radicals undergo methide and hydride shift just like carbonium ion intermediate. C. Radicals are electrophilic species. D. Radicals can be stabilized by both electron-donating and electron-withdrawing groups. 14. The major product of each of the reactions can be predicted using Markovnikov' Rule EXCEPT A. Hydrohalogenation reaction of alkynes B. Hydrohalogenation reaction of alkenes C. Hydration reaction of alkynes D. Hydrogenation reaction of alkynes 15. Markovnikov's Rule is based on the formation of A. stable product. B. stable radical intermediate. C. stable transition state. D. stable carbonium ion intermediate.arrow_forwardWhich alkene has (E) configuration? a. b. C. d.arrow_forward
- 149) Why is a tertiary alcohol more reactive with hydrogen halides, than secondary alcohols? Because secondary alcohols are smaller and therefore less reactive for the attack of halide nucleophile. Because tertiary alcohols have more hydrogen atoms available for substitution reactions. Because a tertiary alcohol can form a more stable carbocation as an intermediate in the reaction. Only the 1st and the 2nd statements are acceptable.arrow_forwardIntramolecular reactions are also observed in Friedel–Crafts alkylation. Draw the intramolecular alkylation product formed from each of the following reactants.arrow_forwardorg chem question answer with explanation. ³⁹arrow_forward
- Draw the structure(s) of the major organic product(s) of the following reaction. or. Phyrion Ph₂ +arrow_forwardWhat name reaction would result to the formation of the cyclohexene structure below? R .CO̟Et A. Diels-Alder reaction B. Wittig Reaction C. Friedel-Craft reaction D. Claisen Condensationarrow_forwardWhat is the final product of the following reaction? A. B. CI میں NaOH, Cl2 (excess C. D. ہیں CI مل CI OHarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning



