
(a)
Interpretation:
The product formed in the given reaction has to be drawn.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
In a sigmatropic reaction “ one new sigma-bond is formed as another breaks.”
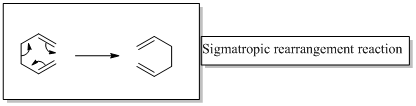
Sigmatropic rearrangement reactions are designated with digits. For example a [1, 3] sigmatropic rearrangement describe a reaction in which the residue migrates from position 1 to position 3.
Migration of carbon and hydrogen will occur in a sigmatropic rearrangement reaction.
(b)
Interpretation:
The product formed in the given reaction has to be drawn.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
In a sigmatropic reaction “ one new sigma-bond is formed as another breaks.”

Sigmatropic rearrangement reactions are designated with digits. For example a [1, 3] sigmatropic rearrangement describe a reaction in which the residue migrates from position 1 to position 3.
Migration of carbon and hydrogen will occur in a sigmatropic rearrangement reaction.
(c)
Interpretation:
The product formed in the given reaction has to be drawn.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
In a sigmatropic reaction “ one new sigma-bond is formed as another breaks.”

Sigmatropic rearrangement reactions are designated with digits. For example a [1, 3] sigmatropic rearrangement describe a reaction in which the residue migrates from position 1 to position 3.
Migration of carbon and hydrogen will occur in a sigmatropic rearrangement reaction.
(d)
Interpretation:
The product formed in the given reaction has to be drawn.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
In a sigmatropic reaction “ one new sigma-bond is formed as another breaks.”

Sigmatropic rearrangement reactions are designated with digits. For example a [1, 3] sigmatropic rearrangement describe a reaction in which the residue migrates from position 1 to position 3.
Migration of carbon and hydrogen will occur in a sigmatropic rearrangement reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
- When (R)-6-bromo-2,6-dimethylnonane is dissolved in CH3OH, nucleophilic substitution yields an optically inactive solution. When the isomeric halide (R)-2-bromo-2,5-dimethylnonane is dissolved in CH3OH under the same conditions, nucleophilic substitution forms an optically active solution. Draw the products formed in each reaction, and explain why the difference in optical activity is observed.arrow_forwardPredict the stereochemical outcome of the following E2 reaction: CI CIarrow_forwardProvide the major monobromination product for the following reactionsarrow_forward
- Starting from the enantiomerically pure alkyl chloride, provide a two-step synthesis to arrive at the racemic mixture of epoxides shown below as the major products.arrow_forward9. Draw the product(s) of the following intramolecular reaction and indicate stereochemistry when relevant. Lum LIH intramolecular SN1arrow_forward7B What is the product of the following cleavage reaction and what is its stereochemistry? Me H Br H NaOEtarrow_forward
- 13) Draw the major product of the following reaction: e H¹, H₂Oarrow_forwardtert-Cumyl chloride undergoes an Sy1 solvolysis reaction in aqueous acetone. CH3 CH3 H2O CI OH acetone ČH3 CH3 Rank the derivatives of tert-cumyl chloride in order of increasing reactivity in this solvolysis. least reactive p-methoxy-tert-cumyl chloride m-nitro-tert-cumyl chloride p-fluoro-tert-cumyl chloride p-nitro-tert-cumyl chloride most reactivearrow_forwardIdentify the product of the following two-step reaction sequence. The initial intermediate formed from Step [1] undergoes a [3,3] sigmatropic rearrangement prior to reaction with CH3I.arrow_forward
- 2. What is the E2 elimination product from the following reaction? There is only one. H CI H KOH E2arrow_forwardDraw the product(s) of the following reaction, including all stereoisomers: NBS, A peroxidearrow_forwardWhen (S)-1-bromo-1-phenylethane undergoes an SN2 reaction with methanethiol (CH3SH), the product of the reaction is the compound shown below. What will its configuration be? O S only R only O both R and S with slightly more S than R O both R and S with slightly more R than Sarrow_forward
