
College Physics
1st Edition
ISBN: 9781938168000
Author: Paul Peter Urone, Roger Hinrichs
Publisher: OpenStax College
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 28, Problem 49PE
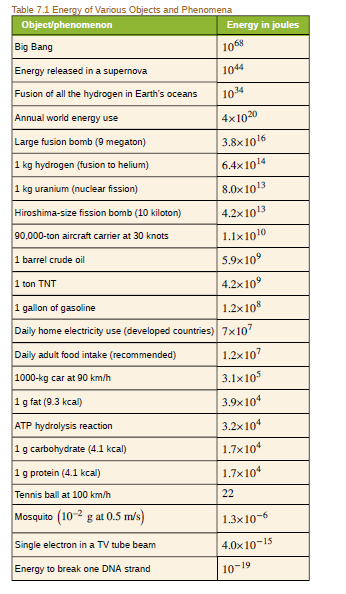
(a) Using data from Table 7.1, calculate the amount of mass converted to energy by the fusion of 1.00 kg of hydrogen. (b) What is the ratio of mass destroyed to the original mass,
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Suppose that you have found a way to convert the rest energy of any type of matter directly to usable energy with an efficiency of 61.0%. How many liters of water would be sufficient fuel to very slowly push the Moon 1.70 mm away from the Earth? The density of water is ?water=1.00kg/liter, the Earth's mass is ?earth=5.97×1024 kg, the Moon's mass is ?moon=7.36×1022 kg, and the separation of the Earth and Moon is ?E,M=3.84×108 m.
What must be the momentum of a particle with mass m so that the total energy of the particle is 3.00 times its rest energy?
How fast must a rocket travel relative to the earth so that time in the rocket "slows down" to quarter its rate as measured by earth-based observers?
Express your answer in meters per second to three significant figures.
Do present-day jet planes approach such speeds? Yes or No
Chapter 28 Solutions
College Physics
Ch. 28 - Which of Einstein's postulates of special...Ch. 28 - Is Earth an inertial frame of reference? Is the...Ch. 28 - When you are flying in a commercial jet, it may...Ch. 28 - Does motion affect the rate of a clock as measured...Ch. 28 - To whom does the elapsed time for a process seem...Ch. 28 - How could you travel far into the future without...Ch. 28 - To does an object seem greater in length, an...Ch. 28 - Relativistic effects such as time dilation and...Ch. 28 - Suppose an astronaut is moving relative to the...Ch. 28 - Explain the meaning of the terms "red shift" and...
Ch. 28 - What happens to the relativistic Doppler effect...Ch. 28 - Is the relativistic Doppler effect consistent with...Ch. 28 - All galaxies farther away than about 50106ly...Ch. 28 - How does modern relativity modify the law of...Ch. 28 - Is it possible for an external force to be acting...Ch. 28 - How are the classical laws of conservation of...Ch. 28 - What happens to the mass of water in a pot when it...Ch. 28 - Consider a thought experiment. You place an...Ch. 28 - The mass of the fuel in a nuclear reactor...Ch. 28 - We know that the velocity of an object with mass...Ch. 28 - Given the fact that light travels at c, can it...Ch. 28 - If you use an Earth-based telescope to project a...Ch. 28 - (a) What is if v=0.250c ? (b) If v=0.500c ?Ch. 28 - (a) What is if v=0.100c ? (b) If v=0.900c ?Ch. 28 - Particles called -mesons are produced by...Ch. 28 - Suppose a particle called a kaon is created by...Ch. 28 - A neutral -meson is a particle that can be...Ch. 28 - A neutron lives 900 s when at rest relative to an...Ch. 28 - If relativistic effects are to be less than 1%,...Ch. 28 - If relativistic effects are to be less than 3%,...Ch. 28 - (a) At what relative velocity is =1.50 ? (b) At...Ch. 28 - (a) At what relative velocity is =2.00 ? (b) At...Ch. 28 - Unreasonable Results (a) Find the value of for...Ch. 28 - A spaceship, 200 m long as seen on board, moves by...Ch. 28 - How fast would a 6.0 m-long sports car have to be...Ch. 28 - (a) How far does the muon in Example 28.1 travel...Ch. 28 - (a) How long would the muon in Example 28.1 have...Ch. 28 - (a) How long does it take the astronaut in Example...Ch. 28 - (a) How fast would an athlete need to be running...Ch. 28 - Unreasonable Results (a) Find the value of for...Ch. 28 - Unreasonable Results A spaceship is heading...Ch. 28 - Suppose a spaceship heading straight towards the...Ch. 28 - Repeat the previous problem with the ship heading...Ch. 28 - If a spaceship is approaching the Earth at 0.100c...Ch. 28 - (a) Suppose the speed of light were only 3000 m/s....Ch. 28 - If a galaxy moving away from the Earth has a speed...Ch. 28 - A space probe speeding towards the nearest star...Ch. 28 - If two spaceships are heading directly towards...Ch. 28 - Two planets are on a collision course, heading...Ch. 28 - When a missile is shot from one spaceship towards...Ch. 28 - What is the relative velocity of two spaceships if...Ch. 28 - Near the center of our galaxy, hydrogen gas is...Ch. 28 - A highway patrol officer uses a device that...Ch. 28 - Prove that for any relative velocity v between two...Ch. 28 - Show that for any relative velocity v between two...Ch. 28 - (a) All but the closest galaxies are receding from...Ch. 28 - Find the momentum of a helium nucleus having a...Ch. 28 - What is the momentum of an electron traveling at...Ch. 28 - (a) Find the momentum of a 1.00109 kg asteroid...Ch. 28 - (a) What is the momentum of a 2000 kg satellite...Ch. 28 - What is the velocity of an electron that has a...Ch. 28 - Find the velocity of a proton that has a momentum...Ch. 28 - (a) Calculate the speed of a 1.00- g particle of...Ch. 28 - (a) Calculate for a proton that has a momentum of...Ch. 28 - What is the rest energy of an electron, given its...Ch. 28 - Find the rest energy in joules and MeV of a...Ch. 28 - If the rest energies of a proton and a neutron...Ch. 28 - The Big Bang that began the universe is estimated...Ch. 28 - A supernova explosion of a 2.001031 kg star...Ch. 28 - (a) Using data from Table 7.1, calculate the mass...Ch. 28 - (a) Using data from Table 7.1, calculate the...Ch. 28 - There is approximately 1034 J of energy available...Ch. 28 - A muon has a rest mass energy of 105.7 MeV, and it...Ch. 28 - A -meson is a particle that decays into a muon...Ch. 28 - (a) Calculate the relativistic kinetic energy of a...Ch. 28 - Alpha decay is nuclear decay in which a helium...Ch. 28 - (a) Beta decay is nuclear decay in which an...Ch. 28 - A positron is an antimatter version of the...Ch. 28 - What is the kinetic energy in MeV of a -meson...Ch. 28 - Find the kinetic energy in MeV of a neutron with a...Ch. 28 - (a) Show that (pc)2/(m c 2)2=21. This means that...Ch. 28 - One cosmic ray neutron has a velocity of 0.250c...Ch. 28 - What is for a proton having a mass energy of...Ch. 28 - (a) What is the effective accelerating potential...Ch. 28 - (a) Using data from Table 7.1, find the mass...Ch. 28 - (a) Calculate the energy released by the...Ch. 28 - A Van de Graaff accelerator utilizes a 50.0 MV...Ch. 28 - Suppose you use an average of 500kWh of electric...Ch. 28 - (a) A nuclear power plant converts energy from...Ch. 28 - Nuclear-powered rockets were researched for some...Ch. 28 - The Sun produces energy at a rate of 4.001026 W by...Ch. 28 - Unreasonable Results A proton has a mass of...Ch. 28 - Construct Your Own Problem Consider a highly...Ch. 28 - Construct Your Own Problem Consider an astronaut...
Additional Science Textbook Solutions
Find more solutions based on key concepts
1. If an object is not moving, does that mean that there are no forces acting on it? Explain.
College Physics: A Strategic Approach (4th Edition)
1. When is energy most evident?
Conceptual Physics (12th Edition)
29. How long would it take you to walk to the moon, end how many steps would you have to take, assuming that yo...
College Physics (10th Edition)
The electromagnetic spectrum of light is often arranged in terms of frequency. Which one of the following has t...
Lecture- Tutorials for Introductory Astronomy
1. An object is subject to two forces that do not point in opposite directions. Is it possible to choose their ...
College Physics: A Strategic Approach (3rd Edition)
66. Astronauts use a centrifuge to simulate the acceleration of a rocket launch. The centrifuge takes 30 s to...
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- In fact, the conversion of mass to energy in the Sun is not 100% efficient. As we have seen in the text, the conversion of four hydrogen atoms to one helium atom results in the conversion of about 0.02862 times the mass of a proton to energy. How much energy in joules does one such reaction produce? (See Appendix E for the mass of the hydrogen atom, which, for all practical purposes, is the mass of a proton.)arrow_forwardOne cosmic ray neuron has a velocity of 0.250c relative to the Earth. (a) What is the neutron's total energy in MeV? (b) Find its momentum. (c) Is in this situation? Discuss in terms of the equation given in part (a) of the previous problem.arrow_forwardThe mass of the fuel in a nuclear reactor decreases by an observable amount as it puts out energy. Is the same true for the coal and oxygen combined in a conventional power plant? If so, is this observable in practice for the coal and oxygen? Explain.arrow_forward
- Find the kinetic energy in MeV of a neutron with a measured life span of 2065 s, given its rest energy is 939.6 MeV, and rest life span is 900s.arrow_forwardIf the rest energies of a proton and a neutron (the two constituents of nuclei) are 938.3 and 939.6 MeV, what is the difference in their mass in kilograms?arrow_forwardCheck Your Understanding Shaw that if a time increment dt elapses for an observer who sees the particle moving with velocity v, it corresponds to a proper lime particle increment for the particle of d=dt.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax
University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax Classical Dynamics of Particles and SystemsPhysicsISBN:9780534408961Author:Stephen T. Thornton, Jerry B. MarionPublisher:Cengage Learning
Classical Dynamics of Particles and SystemsPhysicsISBN:9780534408961Author:Stephen T. Thornton, Jerry B. MarionPublisher:Cengage Learning AstronomyPhysicsISBN:9781938168284Author:Andrew Fraknoi; David Morrison; Sidney C. WolffPublisher:OpenStax
AstronomyPhysicsISBN:9781938168284Author:Andrew Fraknoi; David Morrison; Sidney C. WolffPublisher:OpenStax Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning

University Physics Volume 3
Physics
ISBN:9781938168185
Author:William Moebs, Jeff Sanny
Publisher:OpenStax

Classical Dynamics of Particles and Systems
Physics
ISBN:9780534408961
Author:Stephen T. Thornton, Jerry B. Marion
Publisher:Cengage Learning

Astronomy
Physics
ISBN:9781938168284
Author:Andrew Fraknoi; David Morrison; Sidney C. Wolff
Publisher:OpenStax

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Modern Physics
Physics
ISBN:9781111794378
Author:Raymond A. Serway, Clement J. Moses, Curt A. Moyer
Publisher:Cengage Learning