
(a)
Interpretation: MOs for the given conjugated systems has to be drawn and the HOMO has to be predicted as symmetric or antisymmetric.
Concept introduction:
Conjugated system: A system of connected p-orbitals with delocalized electrons with alternating single and multiple bonds and the compound may be cyclic, linear or mixed.
Molecular orbital theory suggests that atomic orbitals of different atoms combines to create molecular orbitals.
Molecular orbitals can be constructed from linear combination of atomic orbitals.
Bonding orbotals are formed by the additive combination of atomic orbitals and the antibonding orbitals are formed by the substractive combination of atomic orbitals.
Antibonding orbital is a molecular orbital that results when two parallel atomic orbitals with opposite phases interact.
Antibonding orbitals have higher energy than the bonding molecular orbitals.
Ground state and and exited states are the positions with lower and higher energy respectively.
HOMO is a molecular orbital which is the abbrevation of Highest Occupied Molecular Orbital.
LUMO is also a molecular orbital which is the short form of Lowest Unoccupied Molecular Orbital.
If the lobes at the ends of the MO are in phase, then the MO is symmetric.
If the two lobes are out phase then the MO is antisymmetric.
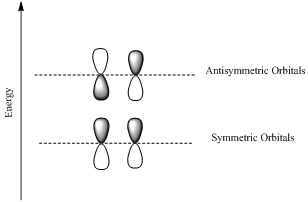
Woodward –Hoffmann rules are the set of rules used to vindicate or predict certain aspects of the stereo chemical outcome and activation energy of pericyclic reactions.
Woodward – Hoffmann rules for Electrocyclic reactions are listed below
(b)
Interpretation: The validity of Woodward – Hoffmann rule for an electrocyclic reaction has to be checked using the MOs of given systems.
Concept introduction:
Woodward –Hoffmann rules are the set of rules used to vindicate or predict certain aspects of the stereo chemical outcome and activation energy of pericyclic reactions.
Woodward – Hoffmann rules for Electrocyclic reactions are listed below
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry (8th Edition)
- Consider the reaction of two compounds ‘A’ and ‘B’ which could make two possible diastereomers ‘AB’ and ‘BA’ (much like this week’s Diels Alder reaction). Hand-write your calculations and responses to the following questions and upload your work as a .jpg or .pdf file. Which of the two products (A or B) will form in greater abundance under thermodynamic control? Which will form in greater abundance under kinetic control? Explain your responses using a sketch of the reaction coordinate diagram for the reactions described above.arrow_forwardIs the following true or false? To catalyse a Diels-Alder reaction a common approach involves the use of a Lewis acid capable of lowering the LUMO of the dienophile.arrow_forwardIn the Diels-Alder experiment, the solution containing the desired cycloadduct product is to be cooled slowly. Briefly explain the reason for cooling the solution slowlyarrow_forward
- ) Draw and label the endo and exo isomers of the Diels–Alder adduct of furan and maleimide.(b) Which isomer of the product would you usually expect from this reaction? Explain why this isomer is usually favoredarrow_forwardHow do you rationalize the fact that the cyclohexane A value for phenyl (2.8) is bigger than that for isopropyl (2.21)?arrow_forward. Discuss the truth of the following statement. Explain why it is true or false Every SN1 reaction produces racemic mixtures in the productsarrow_forward
- Write down the Miller index in the grid direction that may be placed on the grid plane (112) among the <110> aromatic groups.arrow_forwardPlease explian the reasoning and priorities when ranking dienes and dienophiles, thank youarrow_forwardHow would one do this using the Felkin-Anh model? also, is the meso or racemic mixture the major product?arrow_forward
- Uemura and coworkers studied a time dependent Diels-Alder reaction which first formed the endo product as the major organic product and with time produced the exo product (J. Org. Chem. 2018, 83, 9300−9304). Show the endo and exo product for the reaction below. Which is the thermodynamic product and which is the kinetic product? Explain your reasoning.arrow_forwardThe pKa of cyclopentane is > 60, which is about what is expected for a hydrogen that is bonded to an sp3 carbon. Explain why cyclopentadiene is a much stronger acid (pKa of 15) even though it too involves the loss of a proton from an sp3 carbon.arrow_forwardThank you for the answer, I am just wondering that if the double bonds should be considered as chiral centres in this molecule?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


