
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 28.SE, Problem 20MP
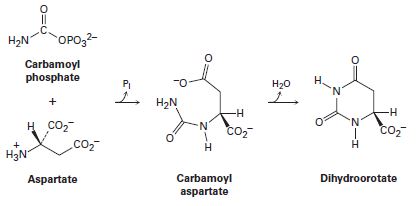
One of the steps in the biosynthesis of uridine monophosphate is the reaction of aspartate with carbamoyl phosphate to give carbamoyl aspartate followed by cyclization to form dihydroorotate. Propose mechanisms for both steps.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
trehalose is a disacharide that can be obtained from fungi sea uchins and insects. acid hydrolysis of trehalose yields only D-glucose. trehalose is hydrolysed by a-glucosidase but not b-glucosidase.methylation of trhalose followed by hydrolysis yield two molar equivalents of 2-3-4-6 -tetra-O-methyl-D-glucopyranose. deduce the structure of the trehalose using the experimental data
The rate constant for the uncatalyzed reaction of two molecules of glycine ethyl ester to form glycylglycine ethyl ester is 0.6 M- 1s - 1. In the presence of
Co2 +, the rate constant is 1.5 * 106 M- 1s - 1. What rate enhancement does the catalyst provide?
The rate constant for the uncatalyzed reaction of two molecules of glycine ethyl ester to form glycylglycine ethyl ester is 0.6 M-1s-1. In the presence of Co2+, the rate constant is 1.5 x 106 M-1s-1. What rate enhancement does the catalyst provide?
Chapter 28 Solutions
Organic Chemistry
Ch. 28.1 - Draw the full structure of the DNA dinucleotide...Ch. 28.1 - Draw the full structure of the RNA dinucleotide...Ch. 28.2 - What sequence of bases on one strand of DNA is...Ch. 28.4 - Show how uracil can form strong hydrogen bonds to...Ch. 28.4 - What RNA base sequence is complementary to the...Ch. 28.4 - From what DNA base sequence was the following RNA...Ch. 28.5 - List codon sequences for the following amino...Ch. 28.5 - List anticodon sequences on the tRNAs carrying the...Ch. 28.5 - What amino acid sequence is coded by the following...Ch. 28.5 - What is the base sequence in the original DNA...
Ch. 28.7 - Prob. 11PCh. 28.7 - Prob. 12PCh. 28.SE - Identify the following bases, and tell whether...Ch. 28.SE - Identify the following nucleotide, and tell how it...Ch. 28.SE - Amine bases in nucleic acids can react with...Ch. 28.SE - The final step in DNA synthesis is deprotection by...Ch. 28.SE - The final step in the metabolic degradation of...Ch. 28.SE - One of the steps in the biosynthesis of a...Ch. 28.SE - One of the steps in the metabolic degradation of...Ch. 28.SE - One of the steps in the biosynthesis of uridine...Ch. 28.SE - Human brain natriuretic peptide (BNP) is a small...Ch. 28.SE - Human and horse insulin both have two polypeptide...Ch. 28.SE - The DNA of sea urchins contains about 32% A. What...Ch. 28.SE - The codon UAA stops protein synthesis. Why does...Ch. 28.SE - Which of the following base sequences would most...Ch. 28.SE - For what amino acids do the following...Ch. 28.SE - Prob. 27APCh. 28.SE - Prob. 28APCh. 28.SE - Draw the complete structure of the ribonucleotide...Ch. 28.SE - Draw the complete structure of the...Ch. 28.SE - Give an mRNA sequence that will code for the...Ch. 28.SE - Give an mRNA sequence that will code for the...Ch. 28.SE - What amino acid sequence is coded for by the...Ch. 28.SE - What amino acid sequence is coded for by the...Ch. 28.SE - Prob. 35APCh. 28.SE - Show the steps involved in a laboratory synthesis...Ch. 28.SE - Draw the structure of cyclic adenosine...Ch. 28.SE - Prob. 38AP
Additional Science Textbook Solutions
Find more solutions based on key concepts
What is the pH range for acidic solutions? For basic solutions?
EBK INTRODUCTION TO CHEMISTRY
Determine the de Brogue wavelength of a. an electron moving at 1/10 the speed of light. b. a 400 g Frisbee movi...
Inorganic Chemistry
Determine the number of protons, neutrons, and electrons in the following atoms: a. a hydrogen atom that has a ...
General, Organic, and Biological Chemistry (3rd Edition)
What is the pH range for acidic solutions? For basic solutions?
Introduction to Chemistry
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living By Chemistry: First Edition Textbook
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In dilute aqueous base, (R)-glyceraldehyde is converted into an equilibrium mixture of (R,S)-glyceraldehyde and dihydroxyacetone. Propose a mechanism for this isomerization.arrow_forwardPropose a mechanism for the biosynthesis of the sesquiterpenoid trichodiene from farnesyl diphosphate. The process involves cyclization to give an intermediate secondary carbocation, followed by several carbocation rearrangements.arrow_forwardThe first step in the metabolism of glycerol, formed by digestion of fats, is phosphorylation of the pro-R—CH2OH group by reaction with adenosine triphosphate (ATP) to give the corresponding glycerol phosphate plus adenosine diphosphate (ADP). Show the stereochemistry of the product.arrow_forward
- Naturally occurring compounds called cyanogenic glycosides, such as lotaustralin, release hydrogen cyanide, HCN, when treated with aqueous acid. The reaction occurs by hydrolysis of the acetal linkage to form a cyanohydrin, which then expels HCN and gives a carbonyl compound. (a) Show the mechanism of the acetal hydrolysis and the structure of the cyanohydrin that results. (b) Propose a mechanism for the loss of HCN, and show the structure of the carbonyl compound that forms.arrow_forwardOne of the later steps in glucose biosynthesis is the isomerization of fructose 6-phosphate to glucose 6-phosphate. Propose a mechanism, using acid or base catalysis as needed.arrow_forwardThe following compound has been found to be an inhibitor of penicillinase. The enzyme can be reactivated by hydroxylamine (NH2OH). Propose a mechanism to account for the inhibition and for the reactivation.arrow_forward
- When the gum of the shrub Sterculia setigera is subjected to acidic hydrolysis, one of the water-soluble components of thehydrolysate is found to be tagatose. The following information is known about tagatose:(1) Molecular formula C6H12O6(2) Undergoes mutarotation.(3) Does not react with bromine water.(4) Reduces Tollens reagent to give d-galactonic acid and d-talonic acid.(5) Methylation of tagatose (using excess CH3 I and Ag2O) followed by acidic hydrolysis gives1,3,4,5-tetra-O-methyltagatose.(a) Draw a Fischer projection structure for the open-chain form of tagatose.(b) Draw the most stable conformation of the most stable cyclic hemiacetal form of tagatosearrow_forwardPropose a mechanism for the biosynthesis of alpha-terpineol from geranyl pyrophosphate.arrow_forwardEthanolamine ammonia lyase, a coenzyme B12–requiring enzyme, catalyzes the following reaction. Propose a mechanism for this reaction.arrow_forward
- The formation of Br2 from NBS first involves the reaction of NBS with HBr to form an iminol intermediate and molecular bromine. The intermediate then undergoes acid-catalyzed tautomerism to form succinimide, the byproduct of the reaction. Propose a curved-arrow mechanism for the conversion of NBS into succinimide that also accounts for the formation of Br2.arrow_forward(S)-Glutamic acid is one of the 20 amino acid building blocks of polypeptides and proteins (Chapter 27). Propose a mechanism for the following conversion.arrow_forward-Amino acids can be prepared by the Strecker synthesis, a two-step process in which an aldehyde is treated with ammonium cyanide followed by hydrolysis of the amino nitrile intermediate with aqueous acid. Propose a mechanism for the reaction.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
CBSE Class 12 Chemistry || Polymers || Full Chapter || By Shiksha House; Author: Best for NEET;https://www.youtube.com/watch?v=OxdJlS0xZ0Y;License: Standard YouTube License, CC-BY