
Concept explainers
Interpretation: The other three stereoisomers of l-isoleucine is to be drawn and the stereogenic centers are to be labeled as
Concept introduction: The stereogenic centers with
Answer to Problem 29.1P
The other three structures of l-isoleucine is,
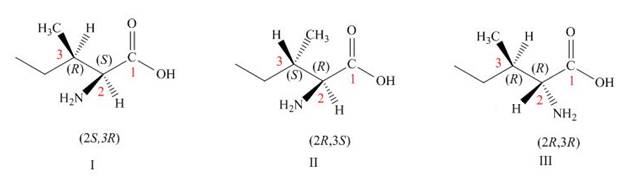
The
Explanation of Solution
The structure of l-isoleucine is,
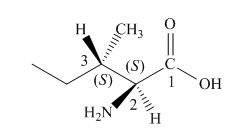
Figure 1
L-leucine has two stereogenic centers.
Maximum number of stereoisomers possible is calculated by the formula,
Where,
• n is the number of stereogenic center.
Substitute the value of stereogenic centres in the above equation.
The
The structures of other stereoisomers are,

Figure 2
The
The other three structures of l-isoleucine is shown in Figure 2. The
Want to see more full solutions like this?
Chapter 29 Solutions
Loose Leaf for Organic Chemistry
- Draw the β anomer of a monosaccharide epimeric with D-glucose at C2 using a Haworth projection.arrow_forwardIsoleucine and threonine are the only two amino acids with two chirality centers. Assign R or S configuration to the methyl-bearing carbon atom of isoleucine.arrow_forwardAssign R or S configuration to each chirality center in the following molecular model of the amino acid isoleucine (blue = N):arrow_forward
- Draw both enantiomers of each amino acid and label them as D or L: (a) methionine; (b) asparaginearrow_forwardDraw Haworth projections for the following: (b) The L isomer of (a)(c) α-D-GlcNAc(d) α-D-Fructofuranosearrow_forwardIdentify the sugar in description. An aldopentose that is not d-arabinose forms d-arabinitol when it is reduced with NaBH4.arrow_forward
- Draw the Haworth projection for the cyclic structure of d-mannose by laying down theFischer projectionarrow_forwardDraw a Fisher Projection of D-Glucose, Haworth Projection of α-D-Glucopyranose and a phosphate ester derivative of glucose.arrow_forwardIdentify the sugar in each description. a. An aldopentose that is not d-arabinose forms d-arabinitol when it is reduced with NaBH4. b. A sugar that is not D-altrose forms d-altraric acid when it is oxidized with nitric acid. c. A ketose that, when reduced with NaBH4, forms d-altritol and d-allitol.arrow_forward
- Draw Fischer projections for the product(s) formed by reaction of d-ribose with the following. In addition, state whether each product is optically active or inactive Q. NaBH4 in H2Oarrow_forwardDraw Fischer projections for the product(s) formed by reaction of d-ribose with the following. In addition, state whether each product is optically active or inactive Q. HNO3, warmarrow_forwardDraw the Fischer projections (D-isomer) of Arabinose and Mannose. Draw the Haworth projections of α-Arabinose and β-Mannose. Draw the structure of β-D-mannopyranosyl-(2→4)-α-D-arabinofuranoside.arrow_forward
