
Concept explainers
(a)
To calculate: The rate at which the x-component changes when the particle moves along the graph of
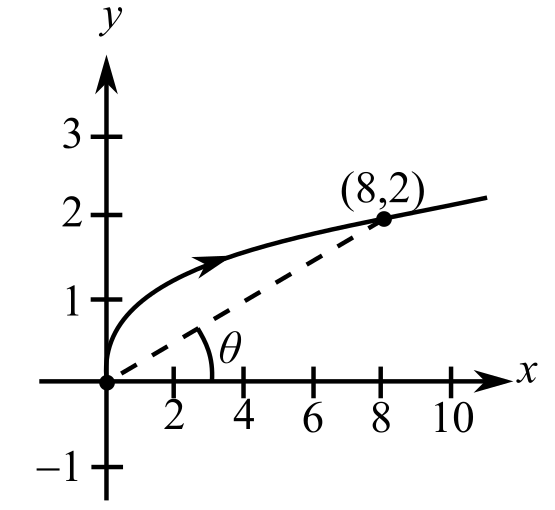
(b)
To calculate: The rate at which the distance from the origin changing at this moment when the particle moves along the graph of
The provided graph is:

(c)
To calculate: The rate at which the angle of inclination
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Calculus: Early Transcendental Functions (MindTap Course List)
- Physics Experiment Consider a physics laboratory experiment designed to determine an unknown mass. A flexible metal meter stick is clamped to a table with 50 centimeters overhanging the edge (see figure). Known masses Mranging from 200grams to 2000grams are attached to the end of the meter stick. For each mass, the meter stick is displaced vertically and then allowed to oscillate. The average time t(in seconds) of one oscillation for each mass is recorded in the table. A model for the data that can be used to predict the time of one oscillation is t=38M+16,96510(M+5000). (a) Use this model to create a table showing the predicted time for each of the masses shown in the table above. (b) Compare the predicted times with the experimental times. What can you conclude? (c) Use the model to approximate the mass of an object for which t=1.056seconds.arrow_forwardVelocity What is the rate of change in directed distance?arrow_forwardVan der Waals Equation In Exercise 18 at the end of Section 2.3, we discussed the ideal gas law, which shows the relationship among volume V, pressure p, and temperature T for a fixed amount 1 mole of a gas. But chemists believe that in many situations, the van der Waals equation gives more accurate results. If we measure temperature T in kelvins, volume V in liters, and pressure p in atmosphere 1 atm is the pressure exerted by the atmosphere at sea level, then the relationship for carbon dioxide is given by p=0.082TV0.0433.592V2atm What volume does this equation predict for 1 mole of carbon dioxide at 500 kelvins and 100 atm?Suggestion: Consider volumes ranging from 0.1 to 1 liter.arrow_forward
- Algebra & Trigonometry with Analytic GeometryAlgebraISBN:9781133382119Author:SwokowskiPublisher:Cengage
 Functions and Change: A Modeling Approach to Coll...AlgebraISBN:9781337111348Author:Bruce Crauder, Benny Evans, Alan NoellPublisher:Cengage Learning
Functions and Change: A Modeling Approach to Coll...AlgebraISBN:9781337111348Author:Bruce Crauder, Benny Evans, Alan NoellPublisher:Cengage Learning College AlgebraAlgebraISBN:9781305115545Author:James Stewart, Lothar Redlin, Saleem WatsonPublisher:Cengage Learning
College AlgebraAlgebraISBN:9781305115545Author:James Stewart, Lothar Redlin, Saleem WatsonPublisher:Cengage Learning  Trigonometry (MindTap Course List)TrigonometryISBN:9781305652224Author:Charles P. McKeague, Mark D. TurnerPublisher:Cengage Learning
Trigonometry (MindTap Course List)TrigonometryISBN:9781305652224Author:Charles P. McKeague, Mark D. TurnerPublisher:Cengage Learning Trigonometry (MindTap Course List)TrigonometryISBN:9781337278461Author:Ron LarsonPublisher:Cengage Learning
Trigonometry (MindTap Course List)TrigonometryISBN:9781337278461Author:Ron LarsonPublisher:Cengage Learning





