
Concept explainers
(a)
Interpretation: Primary, secondary and tertiary hydrogen in below structure should be labeled.
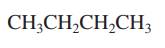
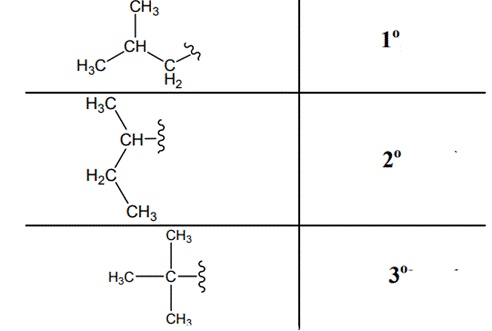
Concept introduction: Primary, secondary and tertiary hydrogen are identified as indicated below:
(a)
Explanation of Solution
The carbon linked to one alkyl/carbon while other two
The carbon linked to two alkyl/carbons and one
(b)
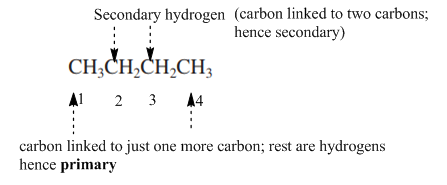
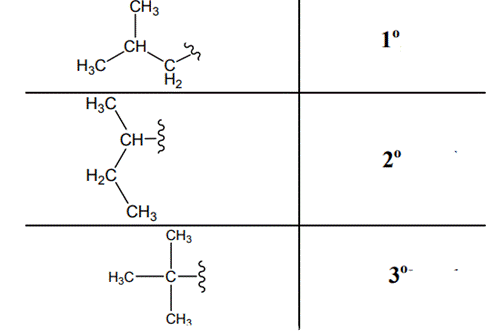
Interpretation: Primary, secondary and tertiary hydrogen in below structure should be labeled.
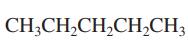
Concept introduction: Primary, secondary and tertiary hydrogen are identified as indicated below:
(b)
Explanation of Solution
The carbon linked to one alkyl/carbon while other two
The carbon linked to two alkyl/carbons and one
(c)
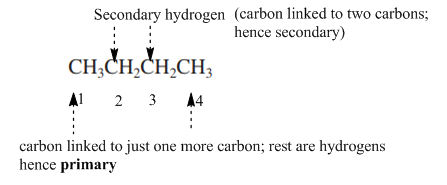
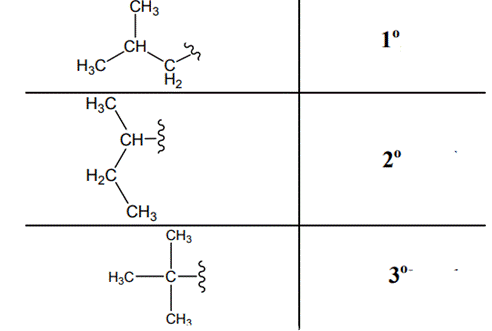
Interpretation: Primary, secondary and tertiary hydrogen in below structure should be labeled.
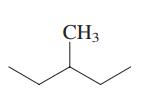
Concept introduction: Primary, secondary and tertiary hydrogen are identified as indicated below:
(c)
Explanation of Solution
The carbon linked to one alkyl/carbon while other two
The carbon linked to two alkyl/carbons and one

(d)
Interpretation: Primary, secondary and tertiary hydrogen in below structure should be labeled.
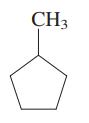
Concept introduction: The carbon linked to one alkyl/carbon while other two
The carbon linked to two alkyl/carbons and one
These are indicated below:
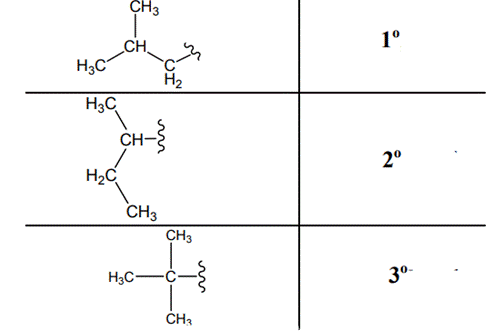
(d)
Explanation of Solution
Since each of the carbon of ring is linked to each other they represent secondary; the methyl group attached to ring represents primary as it is directly linked to only one carbon that is ring carbon. Thus it denotes primary hydrogens. Thus three types of hydrogens are identified and labeled below.
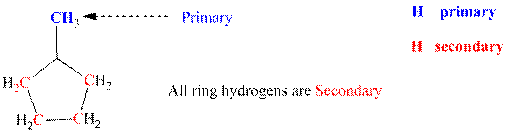
Want to see more full solutions like this?
Chapter 3 Solutions
Organic Chemistry: Structure and Function
- Rank the following groups in order of decreasing priority. a.−F, −NH2, −CH3, −OH b.−CH3, −CH2CH3, −CH2CH2CH3, −(CH2)3CH3 c.−NH2, −CH2NH2, −CH3, −CH2NHCH3 d.−COOH, −CH2OH, −H, −CHO e.−Cl, −CH3, −SH, −OH f.−C≡CH, −CH(CH3)2, −CH2CH3, −CH=CH2arrow_forwardLabel the indicated hydrogen atoms for butane and pentane, as primary (1°), secondary (2°), or tertiary (3°).arrow_forwardHelp with the following question Round the answer to 3 sig figsarrow_forward
- for letter d,e please check if correct D.CH3CH2CH2CH(CH(CH3)2)CH2C(CH2)2(CH3)2CH2CH2OH E.(CH3)2CHOCH2CH3arrow_forwardPlease help with row 2/question 2 ; the instructions for the specific column in row 2 are above row 1: Follow the instructions in each column. Hint for the last column: draw it with a wedge and again with adash – which gives the correct configuration?arrow_forwardtrue or false O2-(g) is more stable than O-(g).arrow_forward
- Which of the following molecules has only single bonds. A. CHCHCH3 B. CH2CHCH3 C. CH3CH2CCH D. CH3CH3 E. CH2CH2 Which of the following molecules has a carbon-to-carbon double bond? A. CH3CCH B. CHCH C. CH3CH3 D. CH3CH2CH3 E. CH2CHCH3arrow_forwardonly need help with PART B. what is the biggest groups? The NH2? The CO2H? the CH3 groups.?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
