
Concept explainers
Interpretation:
The physical states that correspond to the pictorial representation are to be stated.
Concept introduction:
A substance which has some mass and occupies space is termed as matter. It exists in three physical states that are solid, liquid and gas. The force of attraction between atoms determines the
Answer to Problem 1CE
The illustration A, B, C represents the solid state, liquid state and gaseous state respectively.
Explanation of Solution
The given figure is shown below.
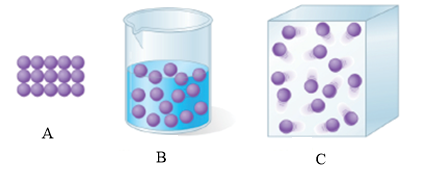
Figure 1
Illustration A is a solid state as the force of attraction between atoms is very strong and the atoms are in a definite shape. On the other hand, illustration B is a liquid state as the atoms are far from each other because the force of attraction between atoms is less. The illustration C represents the gaseous state as atoms are very far from each other because the force of attraction between atoms is negligible.
Illustration A, B, C represents solid state, liquid state and gaseous state respectively.
Want to see more full solutions like this?
Chapter 3 Solutions
Modified Mastering Chemistry with Pearson eText -- Standalone Access Card -- for Introductory Chemistry: Concepts and Critical Thinking (8th Edition)
- Write a brief description of the relationships among each of the following groups of terms or phrases. Answers to the Concept-Linking Exercises are given at the end of the chapter. Homogeneous, heterogeneous, pure substance, mixturearrow_forwardWhich physical state is described as having particles closely packed and held rigidly in position?arrow_forwardWhich of the following is the correct description of theinside of a grapefruit?(a) It is a pure compound.(b) It consists of a homogeneous mixture of compounds.arrow_forward
- Indicate whether each of the following samples of matter is a heterogeneous, homogeneous mixture, element or compound A. A colorless gas, only part of which reacts with hot iron B. A uniform red liquid with a boiling point of 60 degrees C that cannot be broken down into simpler substances using chemical means C. A yellow solid all or which melts at a temperature of 45 degrees C to produce a liquid that decomposes upon further heating D. A single phase liquid that completely evaporates without decomposition when heated to produce a gas that can be seperated into sinpler components using physical meansarrow_forwardA compound is a pure substance whose smallest representative is identified as?arrow_forwardIndicate wether each of the following samples of matter is a heterogenous mixture, homogenous mixture a compound or an element. a) A colorless gas, only part of which reacts with hot ironarrow_forward
- Calcium carbonate (chalk) in not soluble in water. What type of matter results when calcium carbonate is added to water? Classify as an element, compound, homogeneous or heterogeneous mixture.arrow_forwardElemental bromine is a dense, dark red, strong-smelling liquid. Are these characteristics of elemental bromine physical or chemical properties?arrow_forwardState the term that applies to each of the following changes of physical state: (a) Snow changes from a solid to a liquid. (b)Gasoline changes from a liquid to a gas. (c) Dry ice changes from a solid to a gas.arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div




