
Concept explainers
Interpretation:
The leaving group in the given molecule is to be determined inorder identify the positively charged and negatively charged fragments produced during mechanism.
Concept Introduction:
Heterolytic cleavage of sigma bond occurs under a variety of conditions. The mechanism of bond cleavage is as shown below,
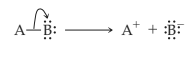
The arrow in the above mechanism indicates that the sigma electrons form A-B bond are leaving A and becoming the exclusive property of B. Since the fragment A is formally losing one electron, it must become positively charged and B must become negatively charged since it gains an electron.
Answer to Problem 1EQ
The mechanism yields a negatively charged Chloride ion and positively charged isopropyl cation.
Explanation of Solution
The given Lewis structure of isopropyl chloride:
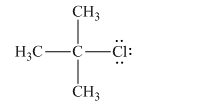
Chlorine is the leaving group in this case. As Chlorine is more electronegative, it obtains the shared electron pair during heterolytic bond cleavage as shown below.
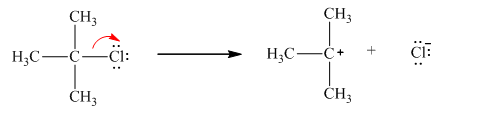
Hence, the cleavage of isopropyl chloride yields chloride ion (negatively charged as shared electron is obtained) and isopropyl cation.
The positive and negative fragments produced in the mechanism are identified.
Want to see more full solutions like this?
Chapter 3 Solutions
Pushing Electrons
- Pick the situation that occurs during radical reaction initiation stage: A. Radicals are formed from nonradicals. B. Nonradicals are formed from other nonradicals C. Nonradicals are formed from radicals D. Radicals are formed from other radicals.arrow_forwardDoes this work? Should there an intermediate step between?arrow_forwardcan yo uplease explain in detail and paragraph the step 2arrow_forward
- Both the Hydroboration and Free Radical addition reactions result in the Anti _________________ product. Complete the sentence above. Explain what it means.arrow_forwardFor 1 and 2, draw the appropriate arrows to show the bond cleavage and bond formations in these reactions. Show the important nonbonding electrons.arrow_forwardGiven the reactant Br−Br, add curved arrows to show homolytic bond cleavage, then draw the expected product. Be sure to add any charges and nonbonding electrons that result from the cleavage.arrow_forward
- Initiation step for (ethane and chlorine)arrow_forwardplease solve this step by step with explainationarrow_forwardCurved arrows are used to illustrate the flow of electrons. Follow the curved arrows and draw the reactant and product of this hydrohalogenation reaction. Include all lone pairs.arrow_forward
- For the reaction below: Draw the major organic structure.arrow_forwarddraw the next 2 steps with the explanationarrow_forwardFill the blank space. Compounds containing a phenol group may work as ANTIOXIDANTS to prevent free radical damage. This is accomplished when a free radical (or UV light) encounters a phenol group, turning the phenol group into a radical. However, contrary to typical radical behavior, the structure of the phenol radical can neutralize (or quench) the unpaired electron. Specifically, the phenol structure neutralizes (or quenches) the unpaired radical electron by doing the following: taking the electron and ---------. The correct name (or abbreviation) of an example compound containing a phenol group with antioxidant properties is: ---------.arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

